AOD-9604
AOD-9604 (Advanced Obesity Drug 9604, hGH Fragment 177-191)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Weight / Metabolic |
| Subcategory | Lipolysis / Fat Metabolism |
| Pharmacological Class | Peptide Fragment |
| Subclass | Human Growth Hormone Fragment (hGH 177-191) |
| Molecular Type | Synthetic Peptide Fragment (15 amino acids corresponding to the C-terminal fragment of human growth hormone, amino acids 177-191, with a tyrosine added at the N-terminus) |
| Origin | Synthetic — derived from the lipolytic domain of human growth hormone |
| Regulatory Status | TGA (Australia's Therapeutic Goods Administration) approved as a food-grade ingredient and as part of an approved osteoarthritis injection (Spamedica). Not FDA-approved. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
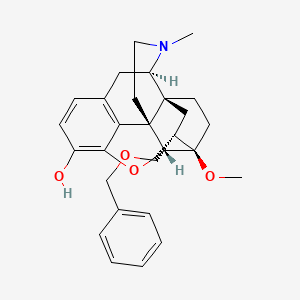
| Molecular Formula | C28H33NO4 |
| Molecular Weight | 447.6 g/mol |
| Exact Mass | 447.24095853 Da |
| InChI Key | LVCBSQDKIDIICL-FRBYVXPPSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of AOD-9604.
Description
AOD-9604 (Advanced Obesity Drug 9604) is a synthetic peptide fragment corresponding to the C-terminal portion of human growth hormone (amino acids 177-191), with an additional tyrosine residue added at the N-terminus for stability. This fragment was identified based on the observation that the lipolytic (fat-burning) activity of growth hormone could be isolated to the C-terminal region of the molecule, separate from the growth-promoting, diabetogenic, and anti-natriuretic effects mediated by other domains. The result is a peptide that reproduces the fat metabolism effects of GH without the growth, IGF-1 elevation, insulin resistance, or fluid retention associated with full-length growth hormone.
The mechanism of action involves stimulation of lipolysis (fat breakdown) and inhibition of lipogenesis (fat synthesis) in adipose tissue. AOD-9604 has been shown to stimulate beta-3 adrenergic receptor-mediated lipolysis, increase fatty acid oxidation, and reduce the activity of lipogenic enzymes in fat cells. Importantly, it does NOT bind to the growth hormone receptor (GHR) — its lipolytic effects are mediated through a distinct, GHR-independent pathway that has not been fully characterized. This GHR independence means that AOD-9604 does not raise IGF-1 levels, does not cause insulin resistance, and does not promote the growth of non-adipose tissues.
In clinical trials conducted primarily in Australia, AOD-9604 administered orally (as a lozenge) produced modest weight loss (approximately 2.6 kg over 12 weeks vs. placebo) in overweight subjects, without significant changes in IGF-1, glucose tolerance, or other growth hormone-related parameters. While the weight loss was statistically significant, it was modest compared to GLP-1 receptor agonists, and the oral development program was not advanced further for obesity. However, AOD-9604 found a different clinical path in Australia — the TGA approved it as a component of an intra-articular osteoarthritis injection based on its demonstrated cartilage repair and anti-inflammatory properties.
Clinical Context
AOD-9604 is positioned as a GH-fragment-based lipolytic agent that provides fat metabolism benefits without the systemic effects of growth hormone. While its weight loss efficacy has been modest in clinical trials, its safety profile is excellent (no GH-like side effects), and its TGA approval in Australia for osteoarthritis provides a regulated clinical precedent. It is commonly used in the peptide space as a complementary agent in body composition protocols, often combined with GH secretagogues, CJC-1295/Ipamorelin, or metabolic peptides.
- Does NOT bind the growth hormone receptor — no GH-like side effects (no IGF-1 elevation, no insulin resistance, no fluid retention, no acromegalic effects)
- TGA approved in Australia for osteoarthritis — regulated precedent
- Lipolytic effects are real but modest compared to GLP-1 receptor agonists
- Well-tolerated with an excellent safety profile — no significant adverse effects in clinical trials
- Does not affect blood glucose or insulin sensitivity — safe in diabetic or pre-diabetic patients
- Can be combined with GH secretagogues without concern for additive GH-related side effects
- The GHR-independent mechanism means it may work through beta-3 adrenergic pathways — not fully elucidated
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to AOD-9604
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Research citations are being compiled for this compound.
Check back soon — our team is curating peer-reviewed sources.
Research Library — 43 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Mixing layer height on the North China Plain and meteorological evidence of serious air pollution in southern Hebei
Xiaowan Zhu, Guiqian Tang, Jianping Guo, et al. · Atmospheric chemistry and physics
Research by Xiaowan Zhu et al., published in Atmospheric chemistry and physics. Not conducted by MedTech Research Group.
Obesity drugs in clinical development.
Jason C. G. Halford · PubMed
Research by Jason C. G. Halford, published in PubMed. Not conducted by MedTech Research Group.
Analytical approaches for the detection of emerging therapeutics and non-approved drugs in human doping controls
Mario Thevis, Wilhelm Schänzer · Journal of Pharmaceutical and Biomedical Analysis
Research by Mario Thevis et al., published in Journal of Pharmaceutical and Biomedical Analysis. Not conducted by MedTech Research Group.
Proteomics in Forensic Analysis: Applications for Human Samples
Van‐An Duong, Jong‐Moon Park, Hee–Joung Lim, et al. · Applied Sciences
Research by Van‐An Duong et al., published in Applied Sciences. Not conducted by MedTech Research Group.
Simplifying and expanding the screening for peptides <2 kDa by direct urine injection, liquid chromatography, and ion mobility mass spectrometry
Andreas Thomas, Christian Görgens, Sven Guddat, et al. · Journal of Separation Science
Research by Andreas Thomas et al., published in Journal of Separation Science. Not conducted by MedTech Research Group.
Progress of Nanomaterials in Photodynamic Therapy Against Tumor
Lei Chen, Jiahui Huang, Xiaotong Li, et al. · Frontiers in Bioengineering and Biotechnology
Research by Lei Chen et al., published in Frontiers in Bioengineering and Biotechnology. Not conducted by MedTech Research Group.
Detecting peptidic drugs, drug candidates and analogs in sports doping: current status and future directions
Mario Thevis, Andreas Thomas, Wilhelm Schänzer · Expert Review of Proteomics
Research by Mario Thevis et al., published in Expert Review of Proteomics. Not conducted by MedTech Research Group.
Analysis of illegal peptide drugs via HILIC-DAD-MS
Steven Janvier, Evelien De Sutter, Evelien Wynendaele, et al. · Talanta
Research by Steven Janvier et al., published in Talanta. Not conducted by MedTech Research Group.
The collaborative study on the genetics of alcoholism: Brain function
Jacquelyn L. Meyers, Sarah J. Brislin, Chella Kamarajan, et al. · Genes Brain & Behavior
Research by Jacquelyn L. Meyers et al., published in Genes Brain & Behavior. Not conducted by MedTech Research Group.
Evaluation of convective storms using spaceborne radars over the <scp>Indo‐Gangetic</scp> Plains and western coast of India
Rohit Mangla, J. Indu, V. Lakshmi · Meteorological Applications
Research by Rohit Mangla et al., published in Meteorological Applications. Not conducted by MedTech Research Group.