ARA-290
ARA-290 (Cibinetide)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Anti-Inflammatory |
| Subcategory | Tissue Protective / Non-Erythropoietic EPO Derivative |
| Pharmacological Class | Peptide |
| Subclass | Innate Repair Receptor (IRR) Agonist / EPO-Derived Tissue Protective Peptide |
| Molecular Type | Synthetic Undecapeptide (11 amino acids) derived from the helix B surface of erythropoietin |
| Origin | Synthetic — designed based on the tissue-protective domain of erythropoietin (EPO), identified by Anthony Bhatt and Michael Brines at Araim Pharmaceuticals |
| Regulatory Status | Investigational. Phase 2 clinical trials conducted for sarcoidosis-associated neuropathy and diabetic neuropathy. Not FDA-approved. |
| Route of Administration | Subcutaneous injection, intravenous (research) |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C51H84N16O21 |
| Molecular Weight | 1257.3 g/mol |
| Exact Mass | 1256.59969375 Da |
| InChI Key | WZTIQQBMSJTRBR-WYKNNRPVSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
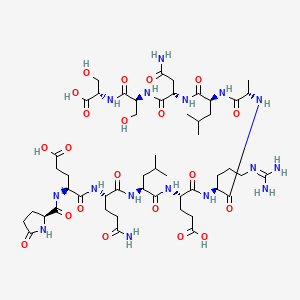
2D structure diagram from NCBI PubChem. This is the actual molecular structure of ARA-290.
Description
ARA-290 (also known as cibinetide) is a synthetic 11-amino-acid peptide derived from the three-dimensional structure of erythropoietin (EPO), specifically from the amino acids that form the exposed surface of helix B (the "B-face") of the EPO molecule. This is a remarkable example of pharmacological precision: EPO is well known to have two distinct biological activities — erythropoiesis (red blood cell production, mediated by the classical homodimeric EPOR/EPOR receptor) and tissue protection/repair (mediated by the heterodimeric Innate Repair Receptor, IRR, composed of EPOR/βcR or EPOR/CD131). ARA-290 was designed to selectively activate only the IRR pathway, providing tissue protective and anti-inflammatory effects without any erythropoietic activity.
The Innate Repair Receptor (IRR) is expressed on a wide variety of cells including neurons, endothelial cells, cardiomyocytes, renal tubular cells, and immune cells (particularly M2-type macrophages). When activated by ARA-290, the IRR triggers JAK2/STAT5 signaling and downstream anti-apoptotic (Bcl-2 upregulation), anti-inflammatory (NF-κB suppression, pro-resolution macrophage polarization), and pro-repair (angiogenesis, Schwann cell migration) pathways. In the context of small fiber neuropathy — a condition characterized by damage to the small unmyelinated C-fibers and thinly myelinated Aδ fibers responsible for pain, temperature, and autonomic function — ARA-290 promotes nerve fiber regeneration by stimulating Schwann cell migration and neurotrophic factor production. Phase 2 clinical trials showed measurable increases in corneal nerve fiber density (a validated biomarker of small fiber neuropathy) and improvements in neuropathic pain scores.
Clinical Context
ARA-290/cibinetide addresses a critical unmet need in neuropathy treatment. Small fiber neuropathy (SFN) affects an estimated 15–20 million people in the United States alone (diabetes is the most common cause, but SFN also occurs in sarcoidosis, autoimmune diseases, and idiopathic cases), and there are currently no FDA-approved treatments that promote nerve fiber regeneration — existing treatments (gabapentin, pregabalin, duloxetine) only manage symptoms. The ability to selectively access tissue-protective EPO signaling without erythropoietic effects (which carry risks of polycythemia, thrombosis, and tumor growth) is a significant pharmacological advance. ARA-290 is in active clinical development.
- Selectively activates the Innate Repair Receptor (IRR) — NO erythropoietic effects (no red blood cell stimulation, no polycythemia risk)
- This is fundamentally different from EPO — ARA-290 does not raise hemoglobin or hematocrit
- Phase 2 trial data available for neuropathy — showed measurable nerve fiber regeneration
- Well-tolerated in clinical trials; most common adverse effects are injection site reactions and mild transient effects
- The tissue-protective mechanism is anti-inflammatory and pro-reparative — it promotes healing, not just symptom management
- Particularly relevant for patients with small fiber neuropathy (SFN), which has no current disease-modifying treatments
- The IRR is widely expressed — tissue-protective effects may extend beyond neurons to cardiac, renal, and vascular tissue
- Not to be confused with EPO or EPO analogs — ARA-290 has no blood-doping or erythropoietic utility
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to ARA-290
12 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
The receptor that tames the innate immune response.
Brines M, Cerami A. Molecular medicine (Cambridge, Mass.), 2012.PMID: 22183892
Erythropoietin and engineered innate repair activators.
Brines M, Cerami A. Methods in molecular biology (Clifton, N.J.), 2013.PMID: 23456859
Erythropoietin receptor (EpoR) agonism is used to treat a wide range of disease.
Sanchis-Gomar F, Perez-Quilis C, Lippi G. Molecular medicine (Cambridge, Mass.), 2013.PMID: 23615965
Joshi D, Abraham D, Shiwen X, Baker D, Tsui J. Journal of vascular surgery, 2014.PMID: 24055514
Swartjes M, van Velzen M, Niesters M, Aarts L, Brines M, et al.. Molecular pain, 2014.PMID: 24529189
Liu Y, Luo B, Han F, Li X, Xiong J, et al.. PloS one, 2014.PMID: 24603865
Collino M, Thiemermann C, Cerami A, Brines M. Pharmacology & therapeutics, 2015.PMID: 25728128
Watanabe M, Lundgren T, Saito Y, Cerami A, Brines M, et al.. Transplantation, 2016.PMID: 26683514
Zhang W, Yu G, Zhang M. Peptides, 2016.PMID: 26774587
Nairz M, Haschka D, Dichtl S, Sonnweber T, Schroll A, et al.. Scientific reports, 2017.PMID: 29026145
The IUPHAR Guide to Immunopharmacology: connecting immunology and pharmacology.
Harding SD, Faccenda E, Southan C, Pawson AJ, Maffia P, et al.. Immunology, 2020.PMID: 32020584
Shokrzadeh M, Etebari M, Ghassemi-Barghi N. Toxicology in vitro : an international journal published in association with BIBRA, 2020.PMID: 32335150
Research Library — 35 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
ERS clinical practice guidelines on treatment of sarcoidosis
Robert P. Baughman, Dominique Valeyre, Peter Korsten, et al. · European Respiratory Journal
Research by Robert P. Baughman et al., published in European Respiratory Journal. Not conducted by MedTech Research Group.
The Many Facets of Erythropoietin Physiologic and Metabolic Response
Sukanya Suresh, Praveen Kumar Rajvanshi, Constance Tom Noguchi · Frontiers in Physiology
Research by Sukanya Suresh et al., published in Frontiers in Physiology. Not conducted by MedTech Research Group.
Erythropoietin and its derivatives: from tissue protection to immune regulation
Bo Peng, Gangcheng Kong, Cheng Yang, et al. · Cell Death and Disease
Research by Bo Peng et al., published in Cell Death and Disease. Not conducted by MedTech Research Group.
Early nerve fibre regeneration in individuals with type 1 diabetes after simultaneous pancreas and kidney transplantation
Shazli Azmi, Maria Jeziorska, Maryam Ferdousi, et al. · Diabetologia
Research by Shazli Azmi et al., published in Diabetologia. Not conducted by MedTech Research Group.
Corneal nerve fiber size adds utility to the diagnosis and assessment of therapeutic response in patients with small fiber neuropathy
Michael Brines, Daniel A. Culver, Maryam Ferdousi, et al. · Scientific Reports
Research by Michael Brines et al., published in Scientific Reports. Not conducted by MedTech Research Group.
Improvements in Diabetic Neuropathy and Nephropathy After Bariatric Surgery: a Prospective Cohort Study
Safwaan Adam, Shazli Azmi, Jan Hoong Ho, et al. · Obesity Surgery
Research by Safwaan Adam et al., published in Obesity Surgery. Not conducted by MedTech Research Group.
Small fiber neuropathy
Mareye Voortman, Daan Fritz, O.J.M. Vogels, et al. · Current Opinion in Pulmonary Medicine
Research by Mareye Voortman et al., published in Current Opinion in Pulmonary Medicine. Not conducted by MedTech Research Group.
Ketamine for pain
Kelly Jonkman, Albert Dahan, Tine van de Donk, et al. · F1000Research
Research by Kelly Jonkman et al., published in F1000Research. Not conducted by MedTech Research Group.
Corneal Confocal Microscopy: A Biomarker for Diabetic Peripheral Neuropathy
Ioannis N. Petropoulos, Georgios Ponirakis, Maryam Ferdousi, et al. · Clinical Therapeutics
Research by Ioannis N. Petropoulos et al., published in Clinical Therapeutics. Not conducted by MedTech Research Group.
Neuropathic Pain: Challenges and Opportunities
Monique van Velzen, Albert Dahan, Marieke Niesters · Frontiers in Pain Research
Research by Monique van Velzen et al., published in Frontiers in Pain Research. Not conducted by MedTech Research Group.