BPC-157
Body Protection Compound-157
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Recovery / Healing |
| Subcategory | Tissue Repair, Cytoprotection, Wound Healing |
| Pharmacological Class | Peptide |
| Subclass | Gastric Pentadecapeptide (cytoprotective agent) |
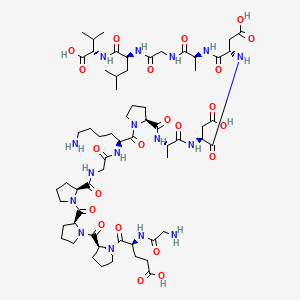
| Molecular Type | Synthetic Peptide (15 amino acids, sequence: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val) |
| Origin | Synthetic fragment derived from human gastric juice protein BPC (Body Protection Compound) |
| Regulatory Status | Research Use Only. Not FDA-approved. No completed human clinical trials (as of 2025). Extensive preclinical data. |
| Route of Administration | Subcutaneous injection (most common for research); also studied via intramuscular, intraperitoneal, and oral routes |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C). Stable in solution at room temperature for short periods. Notably stable in gastric acid (unlike most peptides). |
Chemical Properties
| Molecular Formula | C62H98N16O22 |
| Molecular Weight | 1419.5 g/mol |
| Exact Mass | 1418.70415882 Da |
| InChI Key | HEEWEZGQMLZMFE-RKGINYAYSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of BPC-157.
Description
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide (15 amino acids) derived from a protective protein found in human gastric juice. It is one of the most widely studied peptides in the research peptide market and has generated enormous interest due to its remarkably broad spectrum of protective and healing effects observed in animal models. The parent protein, BPC, is naturally present in gastric juice at very low concentrations and is believed to play a role in the stomach's ability to protect and repair its own mucosal lining — explaining why the stomach, despite being bathed in hydrochloric acid and proteolytic enzymes, maintains its structural integrity.
The mechanism of action of BPC-157 is complex and not fully elucidated, but research has identified several key pathways. BPC-157 upregulates the expression of growth hormone receptors and activates the FAK-paxillin signaling pathway, which is central to cell migration, adhesion, and tissue organization during wound healing. It stimulates angiogenesis (formation of new blood vessels) through upregulation of VEGF (vascular endothelial growth factor) and modulates the nitric oxide (NO) system — both its constitutive and inducible forms. BPC-157 has been shown to interact with the dopaminergic system, protecting against dopamine-related neurotoxicity and modulating dopamine receptor expression. It also interacts with the GABAergic and serotonergic systems, and has demonstrated effects on the opioid system.
In preclinical studies (primarily rat and mouse models), BPC-157 has demonstrated protective and healing effects across a staggering range of tissues and injury types: gastric ulcers, intestinal anastomosis healing, liver damage (including acetaminophen and alcohol-induced hepatotoxicity), tendon and ligament injuries, muscle tears and crush injuries, bone fractures, corneal injuries, peripheral nerve damage (transection and crush), brain injuries, spinal cord injuries, burns, and periodontitis. It has shown cardioprotective effects (arrhythmia prevention, protection against doxorubicin cardiotoxicity), neuroprotective effects (against MPTP-induced Parkinsonism, cuprizone-induced demyelination), and cytoprotective effects against NSAID-induced GI damage. One of its most notable characteristics is its stability in gastric acid, making it one of the rare peptides that maintains biological activity when administered orally — this is believed to relate to its gastric juice origins.
Clinical Context
Despite hundreds of published preclinical studies, BPC-157 has not completed any human clinical trials as of 2025. This creates a significant gap between the extensive animal evidence and clinical applicability. The peptide's popularity in the research and wellness communities stems from anecdotal reports of accelerated recovery from injuries — particularly tendon, ligament, and muscle injuries common in athletes and fitness populations. Its oral bioavailability is a significant practical advantage, though subcutaneous injection near the injury site remains the most common research application approach.
The 20mg vial (YPB.237) offers the best value proposition at $35.10 cost with 80.5% margin at MSRP, and is the preferred size for longer research protocols.
- No completed human clinical trials — all evidence is preclinical (animal models)
- Generally well-tolerated in animal studies with no reported significant adverse effects
- The absence of human safety data means no established contraindications, drug interactions, or adverse effect profiles exist for clinical guidance
- Remarkably stable in acidic environments (pH 2-3), supporting oral administration viability
- Often used in combination with TB500 (Wolverine Blend) for synergistic recovery research
- Multiple routes of administration have been studied; subcutaneous injection near the target tissue is most common for musculoskeletal research
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to BPC-157
8 manually curated + 1 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Stable gastric pentadecapeptide BPC 157: novel therapy in gastrointestinal tract
Sikiric P, Seiwerth S, Rucman R, et al.. Current Pharmaceutical Design, 2011.PMID: 21861828
Comprehensive review of BPC-157's gastroprotective and healing properties across multiple gastrointestinal injury models. Demonstrates cytoprotective effects against NSAIDs, alcohol, and stress-induced ulcers.
BPC 157 and its role in accelerating the healing of transected rat Achilles tendon
Staresinic M, Petrovic I, Novinscak T, et al.. Journal of Orthopaedic Research, 2003.PMID: 12814332
Demonstrated that BPC-157 significantly accelerated Achilles tendon healing in a rat transection model, with improved biomechanical strength and collagen organization compared to controls.
Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts
Chang CH, Tsai WC, Lin MS, et al.. Molecules, 2014.PMID: 25546299
Found that BPC-157 upregulated growth hormone receptor expression in human tendon fibroblasts, suggesting a mechanism for its tendon-healing effects through GH/IGF-1 pathway modulation.
Stable gastric pentadecapeptide BPC 157 in trials for inflammatory bowel disease
Sikiric P, Seiwerth S, Brcic L, et al.. Current Medicinal Chemistry, 2012.PMID: 22300085
Review of BPC-157's efficacy in experimental IBD models including colitis, Crohn's-like lesions, and short bowel syndrome. Demonstrated anti-inflammatory and mucosal healing properties.
Stupnisek M, Franjic S, Drmic D, et al.. Blood Coagulation & Fibrinolysis, 2012.PMID: 22732252
Showed BPC-157 modulates the hemostatic system, reducing bleeding time even in anticoagulated animals, suggesting a unique mechanism of action on vascular homeostasis.
BPC 157 therapy to post-mastectomy lymphedema in a mouse model
Grgic T, Grgic D, Drmic D, et al.. Biomedicines, 2022.PMID: 36140413
Demonstrated BPC-157's ability to reduce lymphedema and promote lymphatic vessel formation in a mastectomy model, expanding its potential applications to lymphatic system repair.
The effect of pentadecapeptide BPC 157 on hippocampal ischemia/reperfusion injuries in rats
Tudor M, Jandric I, Marovic A, et al.. Brain and Behavior, 2010.PMID: 21120131
Showed neuroprotective effects of BPC-157 in a hippocampal ischemia model, with reduced infarct size and improved neuronal survival, supporting its potential in neurodegenerative research.
Perovic D, Kolenc D, Bilic V, et al.. Journal of Orthopaedic Surgery and Research, 2019.PMID: 31337414
BPC-157 improved functional recovery after spinal cord injury in rats and counteracted morphine-induced functional impairment, demonstrating both neuroprotective and neuromodulatory properties.
Jelovac N, Sikiric P, Rucman R, Petek M, Marovic A, et al.. European journal of pharmacology, 1999.PMID: 10499368
2 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
2
Total Trials
1
Recruiting
0
Active
0
Completed
Sponsor: Hudson Biotech · Completed: 2028-02-17
Sponsor: PharmaCotherapia d.o.o. · Completed: 2016-03
Research Library — 939 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
The diverse CB<sub>1</sub>and CB<sub>2</sub>receptor pharmacology of three plant cannabinoids: Δ<sup>9</sup>‐tetrahydrocannabinol, cannabidiol and Δ<sup>9</sup>‐tetrahydrocannabivarin
Roger G. Pertwee · British Journal of Pharmacology
Research by Roger G. Pertwee, published in British Journal of Pharmacology. Not conducted by MedTech Research Group.
Bacterial Degradation of Aromatic Compounds
Jong‐Su Seo, Young-Soo Keum, Qing X. Li · International Journal of Environmental Research and Public Health
Research by Jong‐Su Seo et al., published in International Journal of Environmental Research and Public Health. Not conducted by MedTech Research Group.
Mitochondrial Dysfunction Accounts for the Stochastic Heterogeneity in Telomere-Dependent Senescence
João F. Passos, Gabriele Saretzki, Shaheda Ahmed, et al. · PLoS Biology
Research by João F. Passos et al., published in PLoS Biology. Not conducted by MedTech Research Group.
Definitions of terms relating to mass spectrometry (IUPAC Recommendations 2013)
Kermit K. Murray, Robert K. Boyd, Marcos N. Eberlin, et al. · Pure and Applied Chemistry
Research by Kermit K. Murray et al., published in Pure and Applied Chemistry. Not conducted by MedTech Research Group.
Recommendations for the management of cough in adults
Alyn H. Morice · Thorax
Research by Alyn H. Morice, published in Thorax. Not conducted by MedTech Research Group.
Interleukin-6 and Diabetes
Ole Peter Kristiansen, Thomas Mandrup‐Poulsen · Diabetes
Research by Ole Peter Kristiansen et al., published in Diabetes. Not conducted by MedTech Research Group.
ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI expert consensus recommendations for multimodality imaging in cardiac amyloidosis: Part 1 of 2—evidence base and standardized methods of imaging
Sharmila Dorbala, Yukio Ando, Sabahat Bokhari, et al. · Journal of Nuclear Cardiology
Research by Sharmila Dorbala et al., published in Journal of Nuclear Cardiology. Not conducted by MedTech Research Group.
NAD <sup>+</sup> supplementation normalizes key Alzheimer’s features and DNA damage responses in a new AD mouse model with introduced DNA repair deficiency
Yujun Hou, Sofie Lautrup, Stephanie A. Cordonnier, et al. · Proceedings of the National Academy of Sciences
Research by Yujun Hou et al., published in Proceedings of the National Academy of Sciences. Not conducted by MedTech Research Group.
LSD1/KDM1A inhibitors in clinical trials: advances and prospects
Yuan Fang, Guochao Liao, Bin Yu · Journal of Hematology & Oncology
Research by Yuan Fang et al., published in Journal of Hematology & Oncology. Not conducted by MedTech Research Group.
Effects of cytokines on the liver
Tilo Andus, Joachim Bauer, W. Gerok · Hepatology
Research by Tilo Andus et al., published in Hepatology. Not conducted by MedTech Research Group.