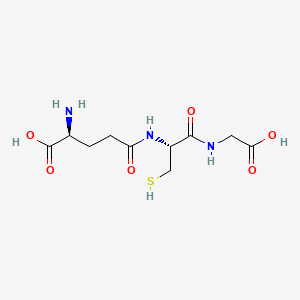
Glutathione
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | GREEN |
| Category | Antioxidants |
| Subcategory | Master Antioxidant / Detoxification |
| Pharmacological Class | Tripeptide |
| Subclass | y-Glutamyl Cysteinyl Glycine (y-peptide bond) |
| Molecular Type | Endogenous Tripeptide (L-Glutamate-L-Cysteine-Glycine, linked by an unusual y-peptide bond between glutamate's y-carboxyl and cysteine's amino group) |
| Origin | Endogenous — synthesized intracellularly by virtually all mammalian cells; the injectable form is exogenous supplementation |
| Regulatory Status | Generally Recognized as Safe (GRAS) as a dietary supplement. Injectable glutathione is used clinically in many countries. Not FDA-approved as a drug for any specific indication. |
| Route of Administration | Intravenous (IV) push or drip, intramuscular injection |
| Reconstitution | Provided in liquid solution (pre-reconstituted); ready for injection |
| Storage | Refrigerate (2-8°C); protect from light |
Chemical Properties
| Molecular Formula | C10H17N3O6S |
| Molecular Weight | 307.33 g/mol |
| Exact Mass | 307.08380644 Da |
| InChI Key | RWSXRVCMGQZWBV-WDSKDSINSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Glutathione.
Description
Glutathione (GSH) is the most abundant intracellular antioxidant in the human body and is arguably the single most important molecule in cellular defense against oxidative stress, electrophilic toxicity, and xenobiotic exposure. It is a tripeptide composed of L-glutamate, L-cysteine, and glycine, with a unique structural feature: the bond between glutamate and cysteine is formed through glutamate's y-carboxyl group (a y-peptide bond) rather than the conventional a-carboxyl group. This y-linkage is critical because it renders glutathione resistant to hydrolysis by most intracellular peptidases (which recognize only a-peptide bonds), allowing it to accumulate to high intracellular concentrations (1-10 mM) — far exceeding typical peptide concentrations.
Glutathione participates in more than 500 enzymatic reactions, functioning as: (1) the primary substrate for glutathione peroxidases (GPx), which reduce hydrogen peroxide and lipid hydroperoxides to water and alcohols, protecting membranes from oxidative damage; (2) the conjugation partner for glutathione S-transferases (GST), which attach glutathione to electrophilic xenobiotics, drugs, and toxins for excretion (Phase II detoxification); (3) a critical cofactor for glyoxalase enzymes that detoxify methylglyoxal (a glycolysis byproduct); (4) a reservoir for cysteine (the rate-limiting amino acid for glutathione synthesis); and (5) a regulator of the cellular redox state (the GSH:GSSG ratio is the primary indicator of cellular oxidative stress). The reduced form (GSH) is oxidized to glutathione disulfide (GSSG) during antioxidant reactions, and GSSG is recycled back to GSH by glutathione reductase using NADPH — maintaining a GSH:GSSG ratio of approximately 100:1 in healthy cells.
Clinical Context
Injectable glutathione is widely used in integrative and functional medicine practices, IV infusion clinics, and wellness centers worldwide. Oral glutathione supplementation has historically been considered ineffective due to rapid degradation in the GI tract by y-glutamyltranspeptidase and peptidases, though liposomal and S-acetyl glutathione formulations have shown improved oral bioavailability. The injectable route bypasses GI degradation entirely, delivering glutathione directly to the bloodstream. Notably, the 600mg and 1500mg vials are priced identically ($35.32), making the 1500mg vial significantly more cost-effective. Glutathione is commonly combined with IV vitamin C, NAD+, and other nutrients in "wellness drip" protocols.
- The 600mg and 1500mg vials are identically priced ($35.32) — the 1500mg vial is the clear value choice
- Injectable glutathione bypasses GI degradation; oral GSH has poor bioavailability
- Administer IV slowly (push over 5-10 minutes) to minimize adverse effects (flushing, mild hypotension)
- The sulfhydryl (thiol) group on cysteine is the functional antioxidant moiety — protect from oxidation during storage
- Glutathione is not stable indefinitely in solution — use promptly after drawing from the vial
- In patients with active malignancy, high-dose antioxidant supplementation is controversial — glutathione could theoretically protect tumor cells from oxidative stress-inducing therapies (chemotherapy, radiation)
- GSH depletion is implicated in a wide range of diseases — supplementation is most beneficial when endogenous levels are demonstrably low (aging, chronic disease, toxic exposure, acetaminophen overdose)
Research data sourced from UniProt. CC BY 4.0 — attribution required.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Biological Function
Catalyzes the production of glutathione from gamma-glutamylcysteine and glycine in an ATP-dependent manner (PubMed:7646467, PubMed:9215686). Glutathione (gamma-glutamylcysteinylglycine, GSH) is the most abundant intracellular thiol in living aerobic cells and is required for numerous processes including the protection of cells against oxidative damage, amino acid transport, the detoxification of foreign compounds, the maintenance of protein sulfhydryl groups in a reduced state and acts as a cofactor for a number of enzymes (PubMed:10369661). Participates in ophthalmate biosynthesis in hepatocytes (By similarity)
Amino acid sequence length: 474 residues
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Glutathione
20 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Srivastava SK, Singhal SS, Hu X, Awasthi YC, Zimniak P, et al.. Archives of biochemistry and biophysics, 1999.PMID: 10334868
CD95-Mediated murine hepatic apoptosis requires an intact glutathione status.
Hentze H, Künstle G, Volbracht C, Ertel W, Wendel A. Hepatology (Baltimore, Md.), 1999.PMID: 10385654
Ritter CA, Bohnenstengel F, Hofmann U, Kroemer HK, Sperker B. Journal of chromatography. B, Biomedical sciences and applications, 1999.PMID: 10437668
Glutathione S-transferase catalyzes the isomerization of (R)-2-hydroxymenthofuran to mintlactones.
Khojasteh-Bakht SC, Nelson SD, Atkins WM. Archives of biochemistry and biophysics, 1999.PMID: 10496977
Tsuboi S. Journal of biochemistry, 1999.PMID: 10544272
Román J, Colell A, Blasco C, Caballeria J, Parés A, et al.. Hepatology (Baltimore, Md.), 1999.PMID: 10573527
Chung KC, Park JH, Kim CH, Lee HW, Sato N, et al.. Journal of neuroscience research, 2000.PMID: 10658192
Soejima A, Ishizuka S, Miyake N, Fukuoka K, Suzuki M, et al.. Experimental nephrology, 2000.PMID: 10729747
The catalytic Tyr-9 of glutathione S-transferase A1-1 controls the dynamics of the C terminus.
Nieslanik BS, Atkins WM. The Journal of biological chemistry, 2000.PMID: 10751412
Galán A, García-Bermejo ML, Troyano A, Vilaboa NE, de Blas E, et al.. The Journal of biological chemistry, 2000.PMID: 10753958
Tolando R, Jovanovic A, Brigelius-Flohé R, Ursini F, Maiorino M. Free radical biology & medicine, 2000.PMID: 10802230
Ligand-induced changes in the structure and dynamics of a human class Mu glutathione S-transferase.
McCallum SA, Hitchens TK, Torborg C, Rule GS. Biochemistry, 2000.PMID: 10858281
Mitochondrial adaptations to obesity-related oxidant stress.
Yang S, Zhu H, Li Y, Lin H, Gabrielson K, et al.. Archives of biochemistry and biophysics, 2000.PMID: 10860543
Wang C, Bammler TK, Guo Y, Kelly EJ, Eaton DL. Toxicological sciences : an official journal of the Society of Toxicology, 2000.PMID: 10869451
Mohamed HE, El-Swefy SE, Hagar HH. Pharmacological research, 2000.PMID: 10887039
Apoptosis induced by extracellular glutathione is mediated by H(2)O(2) production and DNA damage.
Perego P, Gatti L, Carenini N, Dal Bo L, Zunino F. International journal of cancer, 2000.PMID: 10897038
Fahy O, Hammad H, Sénéchal S, Pestel J, Tonnel AB, et al.. American journal of respiratory cell and molecular biology, 2000.PMID: 10919993
A lens glutathione S-transferase, class mu, with thiol-specific antioxidant activity.
Jimenez-Asensio J, Garland D. Experimental eye research, 2000.PMID: 10973735
Arlandson M, Decker T, Roongta VA, Bonilla L, Mayo KH, et al.. The Journal of biological chemistry, 2001.PMID: 11013238
Deng L, Lin-Lee YC, Claret FX, Kuo MT. The Journal of biological chemistry, 2001.PMID: 11020383
50 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
50
Total Trials
1
Recruiting
1
Active
32
Completed
Sponsor: Assiut University · Completed: 2027-04
Sponsor: University of Minnesota · Completed: 2025-12-31
Sponsor: University of Miami · Completed: 2023-03
Sponsor: Stanford University · Completed: 2012-07
Sponsor: Emory University · Completed: 2023-02
Research Library — 240,486 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock, 2012
R.P. Dellinger, Mitchell M. Levy, Andrew Rhodes, et al. · Intensive Care Medicine
Research by R.P. Dellinger et al., published in Intensive Care Medicine. Not conducted by MedTech Research Group.
Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease
Brent R. Stockwell, José Pedro Friedmann Angeli, Hülya Bayır, et al. · Cell
Research by Brent R. Stockwell et al., published in Cell. Not conducted by MedTech Research Group.
Heavy Metal Toxicity and the Environment
Paul B. Tchounwou, Clément G. Yedjou, Anita K. Patlolla, et al. · Proceedings of the Fourth International Symposium on Polarization Phenomena in Nuclear Reactions
Research by Paul B. Tchounwou et al., published in Proceedings of the Fourth International Symposium on Polarization Phenomena in Nuclear Reactions. Not conducted by MedTech Research Group.
Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016
Andrew Rhodes, Laura Evans, Waleed Alhazzani, et al. · Intensive Care Medicine
Research by Andrew Rhodes et al., published in Intensive Care Medicine. Not conducted by MedTech Research Group.
Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018
Lorenzo Galluzzi, Ilio Vitale, Stuart A. Aaronson, et al. · Cell Death and Differentiation
Research by Lorenzo Galluzzi et al., published in Cell Death and Differentiation. Not conducted by MedTech Research Group.
Nitric Oxide and Peroxynitrite in Health and Disease
Pál Pacher, Joseph S. Beckman, Lucas Liaudet · Physiological Reviews
Research by Pál Pacher et al., published in Physiological Reviews. Not conducted by MedTech Research Group.
Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal
Antonio Ayala, Mario Muñoz, Sandro Argüelles · Oxidative Medicine and Cellular Longevity
Research by Antonio Ayala et al., published in Oxidative Medicine and Cellular Longevity. Not conducted by MedTech Research Group.
Biological properties of extracellular vesicles and their physiological functions
Marı́a Yáñez-Mó, Pia Siljander, Zoraida Andreu, et al. · Journal of Extracellular Vesicles
Research by Marı́a Yáñez-Mó et al., published in Journal of Extracellular Vesicles. Not conducted by MedTech Research Group.
Lanthanum Chloride Inhibits LPS Mediated Expressions of Pro-Inflammatory Cytokines and Adhesion Molecules in HUVECs: Involvement of NF-κB-Jmjd3 Signaling
Zhongzhou Chen, Min Xiu, Juanjuan Xing, et al. · Cellular Physiology and Biochemistry
Research by Zhongzhou Chen et al., published in Cellular Physiology and Biochemistry. Not conducted by MedTech Research Group.
Increased oxidative stress in obesity and its impact on metabolic syndrome
Shigetada Furukawa, Takuya Fujita, Michio Shimabukuro, et al. · Journal of Clinical Investigation
Research by Shigetada Furukawa et al., published in Journal of Clinical Investigation. Not conducted by MedTech Research Group.