Hexarelin Acetate
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Growth Hormone Axis |
| Subcategory | Potent Non-Selective GH Secretagogue |
| Pharmacological Class | Peptide |
| Subclass | Growth Hormone Secretagogue / Ghrelin Receptor (GHS-R1a) Agonist |
| Molecular Type | Synthetic Hexapeptide (6 amino acids: His-D-2-MeTrp-Ala-Trp-D-Phe-Lys-NH2) |
| Origin | Synthetic — structural analog of GHRP-6 with 2-methyl substitution on the D-Trp residue |
| Regulatory Status | Research Use Only. Not FDA-approved. Published clinical pharmacology data. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C47H58N12O6 |
| Molecular Weight | 887.0 g/mol |
| Exact Mass | 886.46022762 Da |
| InChI Key | RVWNMGKSNGWLOL-GIIHNPQRSA-N |
| Synonyms |
|
| PubChem | View full record |
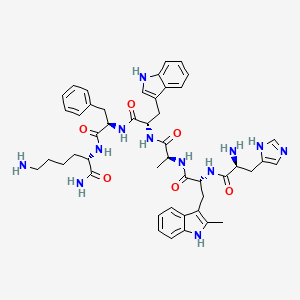
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Hexarelin Acetate.
Description
Hexarelin is the most potent growth hormone secretagogue (GHS) peptide developed to date, producing the largest amplitude GH release pulses of any compound in its class. It is structurally related to GHRP-6 — both are hexapeptides acting on the ghrelin receptor (GHS-R1a) — but hexarelin contains a 2-methyl substitution on the D-tryptophan residue at position 2, which significantly increases its binding affinity and GH-releasing potency. On a milligram-for-milligram basis, hexarelin produces approximately 2-3 times the GH peak amplitude compared to GHRP-6.
However, hexarelin's superior potency comes with a trade-off: it is also the least selective GHS peptide. It produces significant elevations in cortisol and prolactin — more pronounced than GHRP-6 and substantially more than ipamorelin (which produces neither). The cortisol elevation can reach 30-50% above baseline, and prolactin increases are clinically measurable. These effects are dose-dependent and more pronounced at higher doses. Uniquely among GHS peptides, hexarelin also has well-documented cardioprotective properties. Research has demonstrated that hexarelin activates CD36 (cluster of differentiation 36, also known as fatty acid translocase) on cardiac cells, independent of GH release. This CD36 activation has been shown to protect the myocardium against ischemia-reperfusion injury, reduce infarct size, and improve post-ischemic cardiac function in animal models. This cardioprotective mechanism is distinct from GH-mediated effects and represents a unique pharmacological property of hexarelin within the GHS class.
Clinical Context
Hexarelin is typically reserved for situations where maximum GH release potency is desired and the cortisol/prolactin elevations are acceptable or manageable. Its unique cardioprotective properties via CD36 activation have generated significant research interest in cardiology, independent of its GH-releasing effects. A notable limitation is that hexarelin is more susceptible to tachyphylaxis (receptor desensitization) than other GHS compounds — sustained daily use can lead to diminished GH response within 2-4 weeks, necessitating cycling protocols.
- Most potent GHS available but also least selective — significant cortisol and prolactin elevation
- Cortisol elevation of 30-50% above baseline is expected; monitor patients with cortisol-sensitive conditions
- Prolactin elevation may be clinically significant — monitor in patients at risk (gynecomastia, galactorrhea)
- Tachyphylaxis develops more rapidly than with other GHS compounds — cycling is strongly recommended (e.g., 5 days on / 2 days off, or 4 weeks on / 2 weeks off)
- Cardioprotective effects via CD36 are independent of GH release — a unique research application
- Appetite stimulation is present but generally less intense than GHRP-6
- Not recommended as a first-line GH secretagogue due to non-selectivity; ipamorelin or CJC-1295/ipamorelin blend are preferred for standard GH optimization
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Hexarelin Acetate
2 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Hexarelin suppresses high lipid diet and vitamin D3-induced atherosclerosis in the rat.
Pang J, Xu Q, Xu X, Yin H, Xu R, et al.. Peptides, 2010.PMID: 19931584
The effect of hexarelin on growth hormone (GH) secretion in patients with GH deficiency.
Loche S, Cambiaso P, Merola B, Colao A, Faedda A, et al.. The Journal of clinical endocrinology and metabolism, 1995.PMID: 7673411
Research Library — 1,134 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Diagnosis and Complications of Cushing’s Syndrome: A Consensus Statement
Giorgio Arnaldi, Alberto Angeli, A. B. Atkinson, et al. · The Journal of Clinical Endocrinology & Metabolism
Research by Giorgio Arnaldi et al., published in The Journal of Clinical Endocrinology & Metabolism. Not conducted by MedTech Research Group.
Ghrelin Causes Hyperphagia and Obesity in Rats
Alison Wren, Caroline J. Small, Caroline R. Abbott, et al. · Diabetes
Research by Alison Wren et al., published in Diabetes. Not conducted by MedTech Research Group.
Ghrelin
Timo D. Müller, Rubén Nogueiras, Mark L. Andermann, et al. · Molecular Metabolism
Research by Timo D. Müller et al., published in Molecular Metabolism. Not conducted by MedTech Research Group.
Growth Hormone, Insulin-Like Growth Factors, and the Skeleton
Andrea Giustina, Gherardo Mazziotti, Ernesto Canalis · Endocrine Reviews
Research by Andrea Giustina et al., published in Endocrine Reviews. Not conducted by MedTech Research Group.
Ghrelin inhibits leptin- and activation-induced proinflammatory cytokine expression by human monocytes and T cells
Vishwa Deep Dixit, Eric Schaffer, Robert Pyle, et al. · Journal of Clinical Investigation
Research by Vishwa Deep Dixit et al., published in Journal of Clinical Investigation. Not conducted by MedTech Research Group.
Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT
Gianluca Baldanzi, Nicoletta Filigheddu, Santina Cutrupi, et al. · The Journal of Cell Biology
Research by Gianluca Baldanzi et al., published in The Journal of Cell Biology. Not conducted by MedTech Research Group.
Ghrelin, a Natural GH Secretagogue Produced by the Stomach, Induces Hyperglycemia and Reduces Insulin Secretion in Humans
Fabio Broglio, Emanuela Arvat, Andrea Benso, et al. · The Journal of Clinical Endocrinology & Metabolism
Research by Fabio Broglio et al., published in The Journal of Clinical Endocrinology & Metabolism. Not conducted by MedTech Research Group.
Anxiety, Depression, and the Microbiome: A Role for Gut Peptides
Gilliard Lach, Harriët Schellekens, Timothy G. Dinan, et al. · Neurotherapeutics
Research by Gilliard Lach et al., published in Neurotherapeutics. Not conducted by MedTech Research Group.
Endocrine Activities of Ghrelin, a Natural Growth Hormone Secretagogue (GHS), in Humans: Comparison and Interactions with Hexarelin, a Nonnatural Peptidyl GHS, and GH-Releasing Hormone<sup>1</sup>
Emanuela Arvat, Mauro Maccario, Lidia Di Vito, et al. · The Journal of Clinical Endocrinology & Metabolism
Research by Emanuela Arvat et al., published in The Journal of Clinical Endocrinology & Metabolism. Not conducted by MedTech Research Group.
CD36, a scavenger receptor implicated in atherosclerosis
Young Mi Park · Experimental & Molecular Medicine
Research by Young Mi Park, published in Experimental & Molecular Medicine. Not conducted by MedTech Research Group.