Ipamorelin
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Growth Hormone Axis |
| Subcategory | Selective GH Secretagogue |
| Pharmacological Class | Peptide |
| Subclass | Growth Hormone Secretagogue / Ghrelin Receptor (GHS-R1a) Agonist |
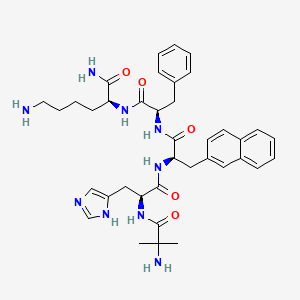
| Molecular Type | Synthetic Pentapeptide (5 amino acids: Aib-His-D-2-Nal-D-Phe-Lys-NH2) |
| Origin | Synthetic — developed by Novo Nordisk, first described in 1998 |
| Regulatory Status | Research Use Only. Not FDA-approved. Extensive clinical trial data available (Novo Nordisk Phase 2). |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C38H49N9O5 |
| Molecular Weight | 711.9 g/mol |
| Exact Mass | 711.38566570 Da |
| InChI Key | NEHWBYHLYZGBNO-BVEPWEIPSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Ipamorelin.
Description
Ipamorelin is the most selective growth hormone secretagogue (GHS) ever developed. It is a synthetic pentapeptide that acts as a potent agonist of the growth hormone secretagogue receptor type 1a (GHS-R1a), commonly known as the ghrelin receptor. What distinguishes ipamorelin from all other GHS compounds — including GHRP-6, GHRP-2, hexarelin, and the natural ligand ghrelin — is its remarkable selectivity: it stimulates GH release from the anterior pituitary without any significant effect on cortisol, prolactin, aldosterone, or appetite. This selectivity is unique in the GHS class and represents a significant pharmacological advancement.
The mechanism of action involves binding to GHS-R1a on pituitary somatotroph cells, activating a Gq-protein coupled signaling cascade that increases intracellular calcium through the phospholipase C/inositol trisphosphate (PLC/IP3) pathway. This calcium rise triggers GH vesicle exocytosis. Unlike GHRP-6, which activates multiple receptor subtypes and downstream pathways (leading to cortisol and prolactin release via hypothalamic CRH and PRL-releasing pathways), ipamorelin's binding profile is highly specific to the GHS-R1a on somatotrophs without significant activation of corticotroph or lactotroph pathways. Additionally, ipamorelin does not stimulate appetite — a notable distinction from ghrelin and GHRP-6, which powerfully drive hunger through vagal afferent and hypothalamic mechanisms. This makes ipamorelin particularly suitable for patients or research subjects where appetite stimulation, cortisol elevation, or prolactin increase would be undesirable.
Clinical Context
Ipamorelin was developed by Novo Nordisk and advanced through Phase 2 clinical trials for post-operative ileus (accelerating return of gastrointestinal function after surgery). While it was not ultimately approved for that indication, the clinical trial data provided extensive human safety and pharmacokinetic information. In the peptide research and clinical space, ipamorelin has become the preferred GH secretagogue for practitioners who want clean, selective GH stimulation. Its outstanding cost efficiency ($26.68 for 10mg) and safety profile make it the most commonly recommended entry point for GH axis research.
- Most selective GHS available — does not raise cortisol, prolactin, or appetite
- No significant hunger stimulation (unlike GHRP-6 and ghrelin)
- Excellent cost-to-efficacy ratio ($26.68/10mg is the lowest cost GH secretagogue in the catalog)
- Requires a functional pituitary gland
- Dose-dependent GH release with a ceiling effect (increasing dose beyond a threshold does not produce additional GH release)
- Well-tolerated with minimal adverse effects; injection site reactions and transient head rush are most commonly reported
- Best administered on an empty stomach; food blunts the response
- Evening dosing preferred to augment nocturnal GH pulse
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Ipamorelin
3 manually curated
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Raun K, Hansen BS, Johansen NL, et al.. European Journal of Endocrinology, 1998.PMID: 9720924
Original characterization of ipamorelin showing selective GH release without affecting cortisol, prolactin, or ACTH — distinguishing it from earlier GH secretagogues.
Safety and efficacy of ipamorelin for postoperative ileus
Greenwood-Van Meerveld B, Tyler K, et al.. Journal of Gastrointestinal Surgery, 2007.PMID: 17436136
Clinical study demonstrating ipamorelin's prokinetic effects on gastrointestinal motility, suggesting applications beyond GH release.
Growth hormone secretagogues: basis for potential clinical applications
Ghigo E, Arvat E, Camanni F.. Growth Hormone & IGF Research, 1998.PMID: 10990145
Review of GH secretagogue mechanisms including ipamorelin's selective ghrelin receptor binding and its implications for clinical growth hormone deficiency treatment.
2 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
2
Total Trials
0
Recruiting
0
Active
2
Completed
Sponsor: Helsinn Therapeutics (U.S.), Inc · Completed: 2009-12
Sponsor: Helsinn Therapeutics (U.S.), Inc · Completed: 2014-05
Research Library — 169 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Strategic Approaches to Optimizing Peptide ADME Properties
Li Di · The AAPS Journal
Research by Li Di, published in The AAPS Journal. Not conducted by MedTech Research Group.
Ghrelin Directly Regulates Bone Formation
Nobuhiro Fukushima, Reiko Hanada, Hitoshi Teranishi, et al. · Journal of Bone and Mineral Research
Research by Nobuhiro Fukushima et al., published in Journal of Bone and Mineral Research. Not conducted by MedTech Research Group.
The co-existence of two growth hormone receptors in teleost fish and their differential signal transduction, tissue distribution and hormonal regulation of expression in seabream
Baowei Jiao, Xigui Huang, Chi Bun Chan, et al. · Journal of Molecular Endocrinology
Research by Baowei Jiao et al., published in Journal of Molecular Endocrinology. Not conducted by MedTech Research Group.
Development of Growth Hormone Secretagogues
Roy G. Smith · Endocrine Reviews
Research by Roy G. Smith, published in Endocrine Reviews. Not conducted by MedTech Research Group.
Systemic ghrelin levels in subjects with growth hormone deficiency are not modified by one year of growth hormone replacement therapy
J. A. M. J. L. Janssen, FM van der Toorn, Leo J. Hofland, et al. · European Journal of Endocrinology
Research by J. A. M. J. L. Janssen et al., published in European Journal of Endocrinology. Not conducted by MedTech Research Group.
The Endogenous Growth Hormone Secretagogue (Ghrelin) Is Synthesized and Secreted by Chondrocytes
Jorge Eduardo Caminos, Oreste Gualillo, Francisca Lago, et al. · Endocrinology
Research by Jorge Eduardo Caminos et al., published in Endocrinology. Not conducted by MedTech Research Group.
Novel and Conventional Receptors for Ghrelin, Desacyl-Ghrelin, and Pharmacologically Related Compounds
Brid Callaghan, John B. Furness · Pharmacological Reviews
Research by Brid Callaghan et al., published in Pharmacological Reviews. Not conducted by MedTech Research Group.
Control of Food Intake by Gastrointestinal Peptides: Mechanisms of Action and Possible Modulation in the Treatment of Obesity
Philip Prinz, Andreas Stengel · Journal of Neurogastroenterology and Motility
Research by Philip Prinz et al., published in Journal of Neurogastroenterology and Motility. Not conducted by MedTech Research Group.
Determination of growth hormone releasing peptides (GHRP) and their major metabolites in human urine for doping controls by means of liquid chromatography mass spectrometry
Andreas Thomas, Sebastian Höppner, Hans Geyer, et al. · Analytical and Bioanalytical Chemistry
Research by Andreas Thomas et al., published in Analytical and Bioanalytical Chemistry. Not conducted by MedTech Research Group.
Specific binding sites for synthetic growth hormone secretagogues in non-tumoral and neoplastic human thyroid tissue
Paola Cassoni, Mauro Papotti, Francesco Catapano, et al. · Journal of Endocrinology
Research by Paola Cassoni et al., published in Journal of Endocrinology. Not conducted by MedTech Research Group.