Mazdutide
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | ORANGE |
| Category | Metabolic / Weight Management |
| Subcategory | Dual-Action Appetite and Glycemic Control |
| Pharmacological Class | Dual Peptide Hormone Analog |
| Subclass | GLP-1 / Glucagon Dual Receptor Agonist |
| Molecular Type | Modified Peptide (oxyntomodulin-based analog) |
| Origin | Synthetic analog of endogenous oxyntomodulin |
| Regulatory Status | Investigational. Developed by Innovent Biologics (China). Phase 3 trials ongoing. Approved in China for obesity (2024). Not FDA-approved in the US. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C207H317N45O65 |
| Molecular Weight | 4476 g/mol |
| Exact Mass | 4475.2950252 Da |
| InChI Key | XRBYWQZGSZWYEJ-HMQIFOERSA-N |
| Synonyms |
|
| PubChem | View full record |
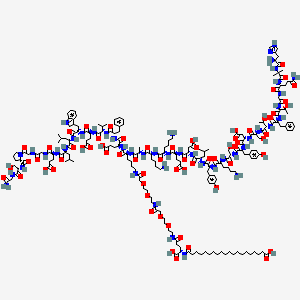
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Mazdutide.
Description
Mazdutide (IBI362, LY3305677) is another dual GLP-1/glucagon receptor agonist, pharmacologically similar to survodutide but developed independently by Innovent Biologics in partnership with Eli Lilly. Like survodutide, it is based on the oxyntomodulin template and activates both GLP-1 and glucagon receptors, combining appetite suppression with increased energy expenditure and hepatic fat metabolism.
Mazdutide has shown clinically significant weight loss in trials conducted primarily in Chinese populations. In Phase 3 GLORY trials, participants achieved approximately 14-17% body weight reduction over 48 weeks. Notably, mazdutide received regulatory approval in China in late 2024 for chronic weight management, making it the first dual GLP-1/glucagon agonist to receive marketing authorization anywhere in the world. The drug is still investigational in the United States and other Western markets.
Clinical Context
The 100mg vial is notably large compared to typical peptide dosing, reflecting the higher per-dose requirements for this compound. The lower margin on this product (27.7% at MSRP) reflects its newer market status and higher manufacturing complexity. Mazdutide occupies a similar therapeutic niche to survodutide and competes with tirzepatide (Zepbound/Mounjaro) and the cagrilintide/semaglutide combination.
- Similar safety profile to survodutide and other GLP-1 agonists
- GI side effects are the most common adverse events
- Less clinical data available in Western populations compared to semaglutide or tirzepatide
- The 100mg vial is designed for multiple doses over several weeks; proper storage and handling are critical
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Mazdutide
5 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Action and therapeutic potential of oxyntomodulin.
Pocai A. Molecular metabolism, 2014.PMID: 24749050
Shankar SS, Shankar RR, Mixson LA, Miller DL, Pramanik B, et al.. Diabetes, 2018.PMID: 29545266
Nalisa DL, Cuboia N, Dyab E, Jackson IL, Felix HJ, et al.. Frontiers in endocrinology, 2024.PMID: 38440786
Shirley M. Drugs, 2025.PMID: 41028652
Approvals by the China NMPA in 2025.
Jing R, Zhi X, Shao L. Nature reviews. Drug discovery, 2026.PMID: 41688808
5 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
5
Total Trials
2
Recruiting
1
Active
0
Completed
Sponsor: Beijing Friendship Hospital · Completed: 2029-09-30
Sponsor: Hangzhou Zhongmei Huadong Pharmaceutical Co., Ltd. · Completed: 2027-12-31
Sponsor: Shanghai Zhongshan Hospital · Completed: 2028-12
Sponsor: Shanghai Zhongshan Hospital · Completed: 2026-06-30
Sponsor: Eli Lilly and Company · Completed: 2026-08
Research Library — 246 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis
Haiqiang Yao, Anqi Zhang, Delong Li, et al. · BMJ
Research by Haiqiang Yao et al., published in BMJ. Not conducted by MedTech Research Group.
What is the pipeline for future medications for obesity?
Eka Melson, Uzma Ashraf, Dimitris Papamargaritis, et al. · International Journal of Obesity
Research by Eka Melson et al., published in International Journal of Obesity. Not conducted by MedTech Research Group.
GLP-1 single, dual, and triple receptor agonists for treating type 2 diabetes and obesity: a narrative review
Nasreen Alfaris, Stephanie Waldrop, Veronica Johnson, et al. · EClinicalMedicine
Research by Nasreen Alfaris et al., published in EClinicalMedicine. Not conducted by MedTech Research Group.
The Pharmaceutical Industry in 2023: An Analysis of FDA Drug Approvals from the Perspective of Molecules
Beatriz G. de la Torre, Fernando Alberício · Molecules
Research by Beatriz G. de la Torre et al., published in Molecules. Not conducted by MedTech Research Group.
An update on peptide-based therapies for type 2 diabetes and obesity
Clifford J. Bailey, Peter R. Flatt, J. Michael Conlon · Peptides
Research by Clifford J. Bailey et al., published in Peptides. Not conducted by MedTech Research Group.
Transforming obesity: The advancement of multi-receptor drugs
Christine M. Kusminski, Diego Pérez–Tilve, Timo D. Müller, et al. · Cell
Research by Christine M. Kusminski et al., published in Cell. Not conducted by MedTech Research Group.
Perioperative management of long-acting glucagon-like peptide-1 (GLP-1) receptor agonists: concerns for delayed gastric emptying and pulmonary aspiration
Mark L. van Zuylen, Sarah E. Siegelaar, Mark P. Plummer, et al. · British Journal of Anaesthesia
Research by Mark L. van Zuylen et al., published in British Journal of Anaesthesia. Not conducted by MedTech Research Group.
A phase 2 randomised controlled trial of mazdutide in Chinese overweight adults or adults with obesity
Linong Ji, Hongwei� Jiang, Zhifeng Cheng, et al. · Nature Communications
Research by Linong Ji et al., published in Nature Communications. Not conducted by MedTech Research Group.
GLP-1 physiology in obesity and development of incretin-based drugs for chronic weight management
Jens J. Holst · Nature Metabolism
Research by Jens J. Holst, published in Nature Metabolism. Not conducted by MedTech Research Group.
Safety and efficacy of a GLP-1 and glucagon receptor dual agonist mazdutide (IBI362) 9 mg and 10 mg in Chinese adults with overweight or obesity: A randomised, placebo-controlled, multiple-ascending-dose phase 1b trial
Linong Ji, Leili Gao, Hongwei� Jiang, et al. · EClinicalMedicine
Research by Linong Ji et al., published in EClinicalMedicine. Not conducted by MedTech Research Group.