Melanotan 2
Melanotan 2 (MT-2)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | ORANGE |
| Category | Skin / Cosmetic |
| Subcategory | Melanogenesis, Tanning |
| Pharmacological Class | Peptide Hormone Analog |
| Subclass | Non-Selective Melanocortin Receptor Agonist |
| Molecular Type | Synthetic Cyclic Heptapeptide (7 amino acids, lactam bridge) |
| Origin | Synthetic — developed at the University of Arizona in the 1980s-1990s as an analog of alpha-MSH (alpha-melanocyte stimulating hormone) |
| Regulatory Status | Research Use Only. Not FDA-approved. Classified as unlicensed in most jurisdictions. Considered a controlled substance in some countries (Australia). |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C). Protect from light. |
Chemical Properties
| Molecular Formula | C26H36N2O5 |
| Molecular Weight | 456.6 g/mol |
| Exact Mass | 456.26242225 Da |
| InChI Key | DLVULHGJZIGXDH-JFLMPSFJSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
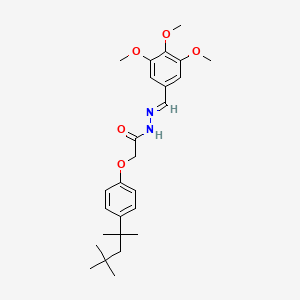
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Melanotan 2.
Description
Melanotan 2 (MT-2) is a synthetic cyclic analog of alpha-melanocyte stimulating hormone (alpha-MSH), a 13-amino-acid endogenous peptide that regulates melanin production in the skin. MT-2 was developed at the University of Arizona by Dr. Victor Hruby and colleagues, who were researching methods to induce protective skin tanning without UV exposure — the original goal was skin cancer prevention through increased melanin production.
MT-2 acts as a non-selective agonist at melanocortin receptors, particularly MC1R (melanocortin 1 receptor, expressed on melanocytes), MC3R, MC4R, and MC5R. Activation of MC1R on melanocytes stimulates eumelanin synthesis (the brown/black protective pigment) through upregulation of the enzyme tyrosinase and other melanogenic enzymes. This increased melanin production darkens the skin, providing enhanced UV protection. However, because MT-2 is non-selective across melanocortin receptor subtypes, it also activates MC4R in the hypothalamus (affecting appetite suppression and sexual arousal) and MC3R (involved in energy homeostasis), leading to several "off-target" effects that have become of independent research interest.
The most notable secondary effect is on sexual function. MC4R activation in the paraventricular nucleus of the hypothalamus triggers erectile responses in males and increased sexual desire in both sexes. This discovery led to the development of PT-141 (bremelanotide), a closely related melanocortin analog specifically optimized for sexual dysfunction (see PT-141 section). MT-2 also suppresses appetite via central MC4R/MC3R activation, has been shown to reduce fat mass in animal studies, and may have cardiovascular effects through peripheral melanocortin receptor activation.
Clinical Context
MT-2 has been widely used in the underground tanning community for years, particularly in Australia, the UK, and Scandinavia, where fair-skinned individuals seek sunless tanning. It has never been approved by any regulatory agency and remains controversial due to potential safety concerns — particularly around melanoma risk (increased melanocyte activity could theoretically promote melanocytic neoplasms, though no causal link has been established in humans), and uncontrolled melanogenesis leading to uneven pigmentation or darkening of existing nevi (moles).
- NOT FDA-approved and considered unlicensed/illegal for human use in many jurisdictions
- Potential melanoma risk is unresolved — no causal evidence, but theoretical concern exists due to melanocyte stimulation
- Common side effects: nausea (especially initially), facial flushing, fatigue, darkening of existing moles/nevi
- Spontaneous erections in males are a common and well-documented side effect
- Appetite suppression may be significant
- New or changing nevi should be evaluated dermatologically
- Contraindicated in patients with history of melanoma or dysplastic nevus syndrome
- Quality control is critical — non-pharmaceutical-grade MT-2 has been associated with adverse events
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Melanotan 2
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Research citations are being compiled for this compound.
Check back soon — our team is curating peer-reviewed sources.
1 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
1
Total Trials
1
Recruiting
0
Active
0
Completed
Sponsor: Hudson Biotech · Completed: 2028-02-17
Research Library — 838 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Glucagon-like peptide 1 (GLP-1)
Timo D. Müller, Brian Finan, Stephen R. Bloom, et al. · Molecular Metabolism
Research by Timo D. Müller et al., published in Molecular Metabolism. Not conducted by MedTech Research Group.
Cancer Treatment Using Peptides: Current Therapies and Future Prospects
Jyothi Thundimadathil · Journal of Amino Acids
Research by Jyothi Thundimadathil, published in Journal of Amino Acids. Not conducted by MedTech Research Group.
Sympathetic and sensory innervation of brown adipose tissue
Timothy J. Bartness, Cheryl H. Vaughan, C. Kay Song · International Journal of Obesity
Research by Timothy J. Bartness et al., published in International Journal of Obesity. Not conducted by MedTech Research Group.
The central melanocortin system directly controls peripheral lipid metabolism
Rubén Nogueiras, Petra Wiedmer, Diego Pérez–Tilve, et al. · Journal of Clinical Investigation
Research by Rubén Nogueiras et al., published in Journal of Clinical Investigation. Not conducted by MedTech Research Group.
The role of proopiomelanocortin (POMC) neurones in feeding behaviour
G. W. M. Millington · Nutrition & Metabolism
Research by G. W. M. Millington, published in Nutrition & Metabolism. Not conducted by MedTech Research Group.
The Neuroendocrine Regulation of Food Intake in Fish: A Review of Current Knowledge
Hélène Volkoff · Frontiers in Neuroscience
Research by Hélène Volkoff, published in Frontiers in Neuroscience. Not conducted by MedTech Research Group.
Requirement of Bardet-Biedl syndrome proteins for leptin receptor signaling
Seongjin Seo, Deng‐Fu Guo, Kevin Bugge, et al. · Human Molecular Genetics
Research by Seongjin Seo et al., published in Human Molecular Genetics. Not conducted by MedTech Research Group.
Prevalence of mutations in LEP, LEPR, and MC4R genes in individuals with severe obesity
Barbara Paolini, Paolo Enrico Maltese, Irene Del Ciondolo, et al. · Genetics and Molecular Research
Research by Barbara Paolini et al., published in Genetics and Molecular Research. Not conducted by MedTech Research Group.
Evaluation of a melanocortin-4 receptor (MC4R) agonist (Setmelanotide) in MC4R deficiency
Tinh‐Hai Collet, B. Dubern, Jacek Mokrosiński, et al. · Molecular Metabolism
Research by Tinh‐Hai Collet et al., published in Molecular Metabolism. Not conducted by MedTech Research Group.
Hair follicle immune privilege and its collapse in alopecia areata
Marta Bertolini, Kevin J. McElwee, Amos Gilhar, et al. · Experimental Dermatology
Research by Marta Bertolini et al., published in Experimental Dermatology. Not conducted by MedTech Research Group.