NAD+
Nicotinamide Adenine Dinucleotide
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | GREEN |
| Category | Longevity / Anti-Aging |
| Subcategory | Cellular Energy, DNA Repair, Sirtuin Activation |
| Pharmacological Class | Coenzyme / Cofactor |
| Subclass | Pyridine Nucleotide |
| Molecular Type | Dinucleotide (not a peptide — a small molecule coenzyme composed of two nucleotides joined through phosphate groups) |
| Origin | Endogenous — NAD+ is present in every living cell and is essential for life. Levels decline approximately 50% between ages 40 and 60. |
| Regulatory Status | Available as dietary supplement (oral NMN, NR). Injectable NAD+ is used in clinical settings (IV drip clinics) but is not FDA-approved for any specific indication. |
| Route of Administration | Subcutaneous injection, intravenous infusion, intramuscular injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water or sterile saline |
| Storage | Refrigerate (2-8°C). Sensitive to heat and light. |
Chemical Properties
| Molecular Formula | C21H27N7O14P2 |
| Molecular Weight | 663.4 g/mol |
| Exact Mass | 663.10912256 Da |
| InChI Key | BAWFJGJZGIEFAR-NNYOXOHSSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
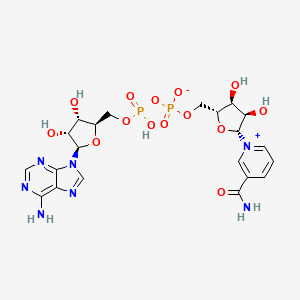
2D structure diagram from NCBI PubChem. This is the actual molecular structure of NAD+.
Description
NAD+ (nicotinamide adenine dinucleotide, oxidized form) is arguably the most important coenzyme in human biology. It participates in over 500 enzymatic reactions and is essential for cellular energy metabolism, DNA repair, gene expression regulation, calcium signaling, and immune function. NAD+ is not a peptide — it is a dinucleotide composed of two nucleotides (nicotinamide mononucleotide and adenosine monophosphate) joined by a phosphodiester bond. Despite not being a peptide, it is included in the research peptide market due to its injectable formulation and its central role in the longevity research space.
NAD+ functions as an electron carrier in metabolic redox reactions, shuttling electrons between enzymes in glycolysis, the citric acid cycle, and oxidative phosphorylation (the mitochondrial electron transport chain). Without adequate NAD+, cells cannot efficiently convert nutrients into ATP (cellular energy). But beyond its metabolic role, NAD+ has become one of the most intensively studied molecules in aging research because of its role as a co-substrate (not just a cofactor — it is consumed, not recycled) for three critical families of enzymes:
-
Sirtuins (SIRT1-7): A family of NAD+-dependent deacetylases and ADP-ribosyltransferases that regulate aging-related processes including DNA repair, inflammatory response, mitochondrial biogenesis, circadian rhythm, and metabolic adaptation. Sirtuins require NAD+ as a co-substrate — when NAD+ levels decline with age, sirtuin activity decreases proportionally, contributing to age-related cellular dysfunction. SIRT1 activation (the "longevity gene" pathway originally linked to caloric restriction benefits) is directly dependent on NAD+ availability.
-
PARPs (Poly-ADP-Ribose Polymerases): Enzymes that consume NAD+ to facilitate DNA repair. PARP1 is the primary DNA damage sensor and repair enzyme in cells. As DNA damage accumulates with age (from oxidative stress, UV, environmental toxins), PARP activity increases, consuming more NAD+ and depleting the pool available for sirtuins — creating a vicious cycle where DNA damage leads to NAD+ depletion which leads to reduced sirtuin activity which leads to further cellular decline.
-
CD38: An ectoenzyme that degrades NAD+ and whose expression increases with age and chronic inflammation. CD38 is now recognized as the primary driver of age-related NAD+ decline — its activity increases 2-3x between young and old tissue, directly consuming the available NAD+ pool.
The age-related decline in NAD+ levels is one of the most well-documented biochemical changes associated with aging. NAD+ levels drop approximately 50% between ages 40 and 60 in human tissues. This decline correlates with (and may directly contribute to) mitochondrial dysfunction, impaired DNA repair capacity, epigenetic drift, stem cell exhaustion, chronic inflammation (inflammaging), and metabolic dysfunction — all hallmarks of the aging process. Restoring NAD+ levels to youthful concentrations has been shown in animal models to reverse many of these aging phenotypes, including improving mitochondrial function, enhancing stem cell regeneration, improving cognitive function, extending lifespan (in mice and worms), and restoring metabolic flexibility.
Clinical Context
NAD+ IV infusion clinics have proliferated in major cities worldwide, offering 250mg-1000mg IV NAD+ drips for anti-aging, energy, addiction recovery, and cognitive enhancement. These clinics typically charge $500-$1,500 per infusion session. The injectable form (subcutaneous or IM) offers a more accessible and affordable delivery method than IV infusion.
The Standard Catalog YPB.101 (Biofermented, 5ml Vial) represents a different formulation — a pre-dissolved liquid form produced by biofermentation rather than chemical synthesis, potentially offering different bioavailability characteristics.
Oral NAD+ precursors (NMN — nicotinamide mononucleotide, and NR — nicotinamide riboside) are widely available as dietary supplements but have lower bioavailability than injectable NAD+ due to first-pass hepatic metabolism and limited intestinal absorption.
- IV infusion can cause chest tightness, nausea, cramping, and a sensation of warmth — these are common and self-limiting; slowing the infusion rate typically resolves symptoms
- Subcutaneous injection may cause local site reactions (burning, stinging)
- NAD+ is a potent vasodilator — monitor blood pressure in hypertensive patients
- Theoretical concern: PARP inhibitors are used in cancer therapy; NAD+ supplementation could theoretically interfere with PARP inhibitor efficacy by restoring the NAD+ pool these drugs aim to deplete
- No significant drug interactions established for injectable NAD+
- The 1000mg PLDS vial (YPB.224) at $75.05 is substantially cheaper than the Biofermented version (YPB.101) at $138.00
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to NAD+
4 manually curated + 16 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
NAD+ metabolism and its roles in cellular processes during ageing
Covarrubias AJ, Perrone R, Grozio A, Verdin E.. Nature Reviews Molecular Cell Biology, 2021.PMID: 33353981
Comprehensive review of NAD+ decline during aging and its role in cellular senescence, inflammation, and metabolic dysfunction. Covers NMN and NR supplementation strategies.
NAD+ intermediates: the biology and therapeutic potential of NMN and NR
Yoshino J, Baur JA, Imai S.. Cell Metabolism, 2018.PMID: 29514064
Review of NAD+ precursors NMN and NR, their pharmacokinetics, tissue distribution, and therapeutic potential for age-related metabolic disorders.
Martens CR, Denman BA, Mazzo MR, et al.. Nature Communications, 2018.PMID: 29599478
First-in-human clinical trial showing oral NR supplementation safely elevates NAD+ levels in healthy older adults with potential cardiovascular benefits.
Gomes AP, Price NL, Ling AJ, et al.. Cell, 2013.PMID: 24360282
Landmark study showing that NAD+ decline causes age-associated mitochondrial dysfunction through HIF-1α destabilization, reversible with NMN treatment in aged mice.
Immunocyte Ca2+ influx system mediated by LTRPC2.
Sano Y, Inamura K, Miyake A, Mochizuki S, Yokoi H, et al.. Science (New York, N.Y.), 2001.PMID: 11509734
Hara Y, Wakamori M, Ishii M, Maeno E, Nishida M, et al.. Molecular cell, 2002.PMID: 11804595
Prosise GL, Luecke H. Journal of molecular biology, 2003.PMID: 12559919
Heiner I, Eisfeld J, Halaszovich CR, Wehage E, Jüngling E, et al.. The Biochemical journal, 2003.PMID: 12564954
Umland TC, Wolff EC, Park MH, Davies DR. The Journal of biological chemistry, 2004.PMID: 15100216
Human SirT1 interacts with histone H1 and promotes formation of facultative heterochromatin.
Vaquero A, Scher M, Lee D, Erdjument-Bromage H, Tempst P, et al.. Molecular cell, 2004.PMID: 15469825
Protasevich II, Brouillette CG, Snow ME, Dunham S, Rubin JR, et al.. Biochemistry, 2004.PMID: 15491144
A candidate NAD+ transporter in an intracellular bacterial symbiont related to Chlamydiae.
Haferkamp I, Schmitz-Esser S, Linka N, Urbany C, Collingro A, et al.. Nature, 2004.PMID: 15577910
Synthesis and antiviral evaluation of cis-substituted cyclohexenyl and cyclohexanyl nucleosides.
Barral K, Courcambeck J, Pèpe G, Balzarini J, Neyts J, et al.. Journal of medicinal chemistry, 2005.PMID: 15658858
Yu M, Schreek S, Cerni C, Schamberger C, Lesniewicz K, et al.. Oncogene, 2005.PMID: 15674325
Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1.
Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, et al.. Nature, 2005.PMID: 15744310
Avalos JL, Bever KM, Wolberger C. Molecular cell, 2005.PMID: 15780941
TRPM2 activation by cyclic ADP-ribose at body temperature is involved in insulin secretion.
Togashi K, Hara Y, Tominaga T, Higashi T, Konishi Y, et al.. The EMBO journal, 2006.PMID: 16601673
Extracellular NAD+ is an agonist of the human P2Y11 purinergic receptor in human granulocytes.
Moreschi I, Bruzzone S, Nicholas RA, Fruscione F, Sturla L, et al.. The Journal of biological chemistry, 2006.PMID: 16926152
Chen L, Wilson DJ, Xu Y, Aldrich CC, Felczak K, et al.. Journal of medicinal chemistry, 2010.PMID: 20491506
Flavins inhibit human cytomegalovirus UL80 protease via disulfide bond formation.
Baum EZ, Ding WD, Siegel MM, Hulmes J, Bebernitz GA, et al.. Biochemistry, 1996.PMID: 8639546
108 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
108
Total Trials
24
Recruiting
6
Active
47
Completed
Sponsor: Mayo Clinic · Completed: 2024-11-08
Sponsor: Maastricht University Medical Center · Completed: 2022-12-07
Sponsor: Société des Produits Nestlé (SPN) · Completed: 2022-03-15
Sponsor: Mayo Clinic · Completed: 2022-12-01
Sponsor: Grupo de Investigación Clínica en Oncología Radioterapia · Completed: 2014-06
Research Library — 23,991 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Nitric Oxide and Peroxynitrite in Health and Disease
Pál Pacher, Joseph S. Beckman, Lucas Liaudet · Physiological Reviews
Research by Pál Pacher et al., published in Physiological Reviews. Not conducted by MedTech Research Group.
Plant Polyphenols as Dietary Antioxidants in Human Health and Disease
Kanti Bhooshan Pandey, Syed Ibrahim Rizvi · Oxidative Medicine and Cellular Longevity
Research by Kanti Bhooshan Pandey et al., published in Oxidative Medicine and Cellular Longevity. Not conducted by MedTech Research Group.
Nitric oxide synthases: regulation and function
Ulrich Förstermann, William C. Sessa · European Heart Journal
Research by Ulrich Förstermann et al., published in European Heart Journal. Not conducted by MedTech Research Group.
Ferroptosis: molecular mechanisms and health implications
Daolin Tang, Xin Chen, Rui Kang, et al. · Cell Research
Research by Daolin Tang et al., published in Cell Research. Not conducted by MedTech Research Group.
Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease
Allison Agus, Julien Planchais, Harry Sokol · Cell Host & Microbe
Research by Allison Agus et al., published in Cell Host & Microbe. Not conducted by MedTech Research Group.
The molecular machinery of regulated cell death
Daolin Tang, Rui Kang, Tom Vanden Berghe, et al. · Cell Research
Research by Daolin Tang et al., published in Cell Research. Not conducted by MedTech Research Group.
Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging
Klaudia Jomová, Renáta Raptová, Suliman Yousef Alomar, et al. · Archives of Toxicology
Research by Klaudia Jomová et al., published in Archives of Toxicology. Not conducted by MedTech Research Group.
Oxidative Stress and Inflammation: What Polyphenols Can Do for Us?
Tarique Hussain, Bie Tan, Yulong Yin, et al. · Oxidative Medicine and Cellular Longevity
Research by Tarique Hussain et al., published in Oxidative Medicine and Cellular Longevity. Not conducted by MedTech Research Group.
Reactive Oxygen Species in Metabolic and Inflammatory Signaling
Steven J. Forrester, Daniel S. Kikuchi, Marina S. Hernandes, et al. · Circulation Research
Research by Steven J. Forrester et al., published in Circulation Research. Not conducted by MedTech Research Group.
Fluorescent chemosensors: the past, present and future
Di Wu, Adam C. Sedgwick, Thorfinnur Gunnlaugsson, et al. · Chemical Society Reviews
Research by Di Wu et al., published in Chemical Society Reviews. Not conducted by MedTech Research Group.