PT-141
PT-141 (Bremelanotide)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is classified as an FDA-regulated compound under the Federal Food, Drug, and Cosmetic Act (FD&C Act), as amended by the Drug Quality and Security Act (DQSA), Title I — Compounding Quality Act (2013).
MedTech Research Group will only fulfill orders for this compound to state-licensed 503A compounding pharmacies or FDA-registered 503B outsourcing facilities that maintain current registration with the U.S. Food and Drug Administration.
Verification Requirements
- Valid state pharmacy license (verified against state board of pharmacy records)
- Current state licensure (503A) or FDA registration (503B) verified via the appropriate state board of pharmacy or FDA Drug Establishment Registration database
- DEA registration (if applicable to the compound)
- Compliance with current Good Manufacturing Practice (cGMP) per 21 CFR Parts 210 and 211
This product may not be sold to consumers, wellness clinics, health stores, or any entity not licensed as a compounding pharmacy. Orders will not be fulfilled until licensure verification is complete. Ref: 21 U.S.C. §§ 353a, 353b; FDA Guidance for Industry: Mixing, Diluting, or Repackaging Biological Products (2025).
| Risk Tier | RED |
| Category | Sexual Health / Fertility |
| Subcategory | Sexual Desire / Arousal |
| Pharmacological Class | Peptide |
| Subclass | Melanocortin-4 Receptor (MC4R) Agonist |
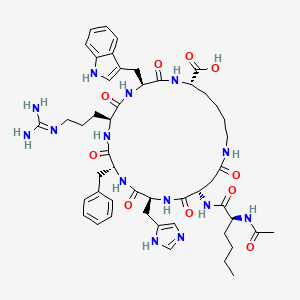
| Molecular Type | Synthetic Cyclic Heptapeptide (7 amino acids, cyclized via lactam bridge: Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH) |
| Origin | Synthetic — derived from Melanotan II; developed by Palatin Technologies |
| Regulatory Status | FDA-approved as Vyleesi (bremelanotide injection) for hypoactive sexual desire disorder (HSDD) in premenopausal women (2019). The first and only FDA-approved peptide for sexual desire. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C50H68N14O10 |
| Molecular Weight | 1025.2 g/mol |
| Exact Mass | 1024.52428442 Da |
| InChI Key | FFHBJDQSGDNCIV-MFVUMRCOSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of PT-141.
Description
PT-141 (bremelanotide) is a synthetic cyclic heptapeptide melanocortin receptor agonist that acts primarily on the melanocortin-4 receptor (MC4R) in the central nervous system to enhance sexual desire and arousal. It represents a mechanistically unique approach to sexual dysfunction — rather than affecting blood flow (like PDE5 inhibitors such as sildenafil/Viagra) or hormonal levels (like testosterone replacement), PT-141 acts directly on the neural pathways in the brain that regulate sexual desire, specifically within the medial preoptic area (mPOA) and paraventricular nucleus (PVN) of the hypothalamus.
PT-141 was derived from Melanotan II (MT-II, also in this catalog), a non-selective melanocortin receptor agonist that was originally developed for tanning but was incidentally found to produce spontaneous penile erections in male research subjects. PT-141 was then specifically engineered as a cyclized, more selective analog focused on the MC4R-mediated sexual response pathway while reducing the MC1R-mediated melanogenic (tanning) activity. The MC4R activation triggers a downstream signaling cascade involving oxytocin release, dopaminergic facilitation, and modulation of nitric oxide synthase in hypothalamic nuclei — collectively producing increased sexual desire, arousal, and genital blood flow through central (brain-mediated) mechanisms rather than peripheral vasodilation.
The FDA approved bremelanotide as Vyleesi in June 2019 for the treatment of acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women — a condition characterized by persistently low or absent sexual desire causing marked distress. This made PT-141 the first centrally-acting peptide approved for sexual desire enhancement and only the second FDA-approved treatment for female HSDD (after flibanserin/Addyi, a 5-HT1A agonist / 5-HT2A antagonist oral medication).
Clinical Context
PT-141 is unique in pharmacology as the only FDA-approved drug that enhances sexual desire through melanocortin receptor activation in the brain. In pivotal trials (RECONNECT studies), bremelanotide significantly increased the number of satisfying sexual events and reduced distress associated with low sexual desire compared to placebo. The approved dosing is 1.75mg subcutaneously, as needed, at least 45 minutes before anticipated sexual activity, with no more than one dose per 24 hours and no more than 8 doses per month. The mechanism is central (brain-based), not peripheral (vascular) — this is a critical distinction from PDE5 inhibitors.
- FDA-approved as Vyleesi for premenopausal female HSDD — extensive safety data
- Centrally acting (brain-based sexual desire) — NOT a vasodilator like sildenafil
- Most common adverse effects: nausea (40% — most common reason for discontinuation), flushing (20%), headache (11%), injection site reactions
- Nausea can be significant — advise patients; anti-emetics may be considered
- Transient blood pressure elevation (~6 mmHg systolic) — use with caution in poorly controlled hypertension or cardiovascular disease
- May cause skin hyperpigmentation with repeated use (residual MC1R activity, though less than Melanotan II)
- Contraindicated in patients with uncontrolled hypertension or known cardiovascular disease
- Two SKUs available: YPB.274 (Premier, $26.33/10mg) and YPB.110 (Standard, $46.00/5mg) — the Premier 10mg vial is significantly more cost-effective
- NOT a daily medication — on-demand use, maximum 8 doses per month
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to PT-141
6 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
PT-141: a melanocortin agonist for the treatment of sexual dysfunction.
Molinoff PB, Shadiack AM, Earle D, Diamond LE, Quon CY. Annals of the New York Academy of Sciences, 2003.PMID: 12851303
Conde-Frieboes K, Thøgersen H, Lau JF, Sensfuss U, Hansen TK, et al.. Journal of medicinal chemistry, 2012.PMID: 22335602
Clayton AH, Lucas J, DeRogatis LR, Jordan R. Clinical therapeutics, 2017.PMID: 28189361
Both S. Current sexual health reports, 2017.PMID: 29225554
Expert opinion on existing and developing drugs to treat female sexual dysfunction.
Miller MK, Smith JR, Norman JJ, Clayton AH. Expert opinion on emerging drugs, 2018.PMID: 30251897
Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder: Two Randomized Phase 3 Trials.
Kingsberg SA, Clayton AH, Portman D, Williams LA, Krop J, et al.. Obstetrics and gynecology, 2019.PMID: 31599840
10 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
10
Total Trials
0
Recruiting
1
Active
9
Completed
Sponsor: Cosette Pharmaceuticals, Inc. · Completed: 2025-11-15
Sponsor: Palatin Technologies, Inc · Completed: 2012-09
Sponsor: Palatin Technologies, Inc · Completed: 2024-04-26
Sponsor: Kwang Dong Pharmaceutical co., ltd. · Completed: 2023-05-16
Sponsor: Palatin Technologies, Inc · Completed: 2017-06-30
Research Library — 66 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Dopamine and Oxytocin Interactions Underlying Behaviors: Potential Contributions to Behavioral Disorders
Tracey Baskerville, Alison J. Douglas · CNS Neuroscience & Therapeutics
Research by Tracey Baskerville et al., published in CNS Neuroscience & Therapeutics. Not conducted by MedTech Research Group.
Cyclic peptide drugs approved in the last two decades (2001–2021)
Huiya Zhang, Shiyu Chen · RSC Chemical Biology
Research by Huiya Zhang et al., published in RSC Chemical Biology. Not conducted by MedTech Research Group.
Evaluation of a melanocortin-4 receptor (MC4R) agonist (Setmelanotide) in MC4R deficiency
Tinh‐Hai Collet, B. Dubern, Jacek Mokrosiński, et al. · Molecular Metabolism
Research by Tinh‐Hai Collet et al., published in Molecular Metabolism. Not conducted by MedTech Research Group.
Advances in therapeutic peptides targeting G protein-coupled receptors
Anthony P. Davenport, Conor C. G. Scully, Chris de Graaf, et al. · Nature Reviews Drug Discovery
Research by Anthony P. Davenport et al., published in Nature Reviews Drug Discovery. Not conducted by MedTech Research Group.
The International Society for the Study of Women's Sexual Health Process of Care for Management of Hypoactive Sexual Desire Disorder in Women
Anita H. Clayton, Irwin Goldstein, Noel N. Kim, et al. · Mayo Clinic Proceedings
Research by Anita H. Clayton et al., published in Mayo Clinic Proceedings. Not conducted by MedTech Research Group.
RM-493, a Melanocortin-4 Receptor (MC4R) Agonist, Increases Resting Energy Expenditure in Obese Individuals
Kong Y. Chen, Ranganath Muniyappa, Brent S. Abel, et al. · The Journal of Clinical Endocrinology & Metabolism
Research by Kong Y. Chen et al., published in The Journal of Clinical Endocrinology & Metabolism. Not conducted by MedTech Research Group.
Endocrinologic Control of Men’s Sexual Desire and Arousal/Erection
Giovanni Corona, Andrea M. Isidori, Antônio Aversa, et al. · The Journal of Sexual Medicine
Research by Giovanni Corona et al., published in The Journal of Sexual Medicine. Not conducted by MedTech Research Group.
An Effect on the Subjective Sexual Response in Premenopausal Women with Sexual Arousal Disorder by Bremelanotide (PT-141), a Melanocortin Receptor Agonist
Lisa E. Diamond, Dennis Earle, Julia R. Heiman, et al. · The Journal of Sexual Medicine
Research by Lisa E. Diamond et al., published in The Journal of Sexual Medicine. Not conducted by MedTech Research Group.
Melanocortins in the Treatment of Male and Female Sexual Dysfunction
Trevor J. Hallam, Carl Spana, Dennis Earle, et al. · Current Topics in Medicinal Chemistry
Research by Trevor J. Hallam et al., published in Current Topics in Medicinal Chemistry. Not conducted by MedTech Research Group.
Sexual Health after a Breast Cancer Diagnosis: Addressing a Forgotten Aspect of Survivorship
Suneela Vegunta, Carol L. Kuhle, Jennifer A. Vencill, et al. · Journal of Clinical Medicine
Research by Suneela Vegunta et al., published in Journal of Clinical Medicine. Not conducted by MedTech Research Group.