Semaglutide
Semaglutide (GLP-1 S)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is classified as an FDA-regulated compound under the Federal Food, Drug, and Cosmetic Act (FD&C Act), as amended by the Drug Quality and Security Act (DQSA), Title I — Compounding Quality Act (2013).
MedTech Research Group will only fulfill orders for this compound to state-licensed 503A compounding pharmacies or FDA-registered 503B outsourcing facilities that maintain current registration with the U.S. Food and Drug Administration.
Verification Requirements
- Valid state pharmacy license (verified against state board of pharmacy records)
- Current state licensure (503A) or FDA registration (503B) verified via the appropriate state board of pharmacy or FDA Drug Establishment Registration database
- DEA registration (if applicable to the compound)
- Compliance with current Good Manufacturing Practice (cGMP) per 21 CFR Parts 210 and 211
This product may not be sold to consumers, wellness clinics, health stores, or any entity not licensed as a compounding pharmacy. Orders will not be fulfilled until licensure verification is complete. Ref: 21 U.S.C. §§ 353a, 353b; FDA Guidance for Industry: Mixing, Diluting, or Repackaging Biological Products (2025).
| Risk Tier | RED |
| Category | Metabolic / Weight Management |
| Subcategory | Appetite Regulation, Glycemic Control |
| Pharmacological Class | Peptide Hormone Analog |
| Subclass | GLP-1 Receptor Agonist (Incretin Mimetic) |
| Molecular Type | Modified Peptide (acylated GLP-1 analog, 31 amino acids) |
| Origin | Synthetic analog of endogenous GLP-1 (glucagon-like peptide-1) |
| Regulatory Status | FDA-approved as Ozempic (Type 2 diabetes, 2017), Wegovy (chronic weight management, 2021), and Rybelsus (oral, T2D, 2019). Compounded versions are sold for research use. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) before reconstitution. After reconstitution, refrigerate and use within 28 days. |
Chemical Properties
| Molecular Formula | C187H291N45O59 |
| Molecular Weight | 4114 g/mol |
| Exact Mass | 4112.1187318 Da |
| InChI Key | DLSWIYLPEUIQAV-CCUURXOWSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
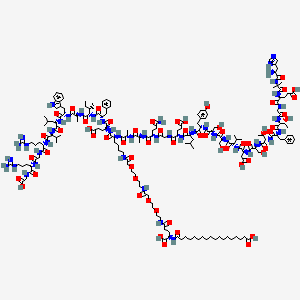
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Semaglutide.
Description
Semaglutide is a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist originally developed by Novo Nordisk. It is a synthetic analog of the naturally occurring incretin hormone GLP-1, which is secreted by L-cells in the small intestine in response to food intake. The molecule has been engineered with specific amino acid substitutions (Aib at position 8, Arg at position 34) and a C-18 fatty diacid chain attached via a linker at position 26, which enables it to bind to albumin in the bloodstream. This albumin binding dramatically extends its half-life to approximately 7 days (compared to the 2-minute half-life of native GLP-1), allowing for once-weekly dosing.
The mechanism of action centers on binding to and activating GLP-1 receptors throughout the body. In the pancreas, this stimulates glucose-dependent insulin secretion from beta cells and suppresses inappropriate glucagon release from alpha cells — meaning it only drives insulin release when blood sugar is elevated, significantly reducing hypoglycemia risk compared to older diabetes medications. In the brain, semaglutide acts on GLP-1 receptors in the hypothalamus and brainstem (particularly the nucleus tractus solitarius and area postrema), reducing appetite and creating a sense of satiety. It also slows gastric emptying, meaning food stays in the stomach longer, further contributing to reduced food intake and improved postprandial glucose levels.
Clinical Context
Semaglutide has become one of the most commercially significant pharmaceutical products in history. The branded injectable versions (Ozempic for diabetes, Wegovy for obesity) have generated billions in revenue and created massive demand that has outstripped supply. Clinical trials demonstrated average weight loss of 14.9% of body weight over 68 weeks in the STEP 1 trial (Wegovy), and an A1C reduction of 1.5-1.8% in the SUSTAIN trials (Ozempic). The SELECT cardiovascular outcomes trial (2023) also demonstrated a 20% reduction in major adverse cardiovascular events (MACE) in overweight/obese adults without diabetes, leading to a cardiovascular indication.
The compounded peptide market has emerged largely because of chronic shortages of branded semaglutide products and the significant cost differential — branded Wegovy lists at approximately $1,350/month, while compounded semaglutide is available at a fraction of that cost. The FDA has allowed compounding of semaglutide during the shortage period, though the regulatory landscape continues to evolve.
- Contraindicated in patients with personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Most common adverse effects: nausea (44%), diarrhea (30%), vomiting (24%) — typically dose-dependent and diminish over time
- Dose titration is essential: start low (0.25mg/week) and increase gradually over 16-20 weeks to minimize GI side effects
- Drug interactions: may affect absorption of oral medications due to delayed gastric emptying
- Pancreatitis risk: rare but reported; discontinue if suspected
- Not recommended during pregnancy or breastfeeding
- Renal: use with caution in patients with renal impairment; dehydration from GI side effects can worsen renal function
- Gallbladder: increased risk of cholelithiasis and cholecystitis
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Semaglutide
4 manually curated + 14 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Once-weekly semaglutide in adults with overweight or obesity (STEP 1)
Wilding JPH, Batterham RL, Calanna S, et al.. New England Journal of Medicine, 2021.PMID: 33567185
Landmark trial demonstrating 14.9% mean body weight loss with semaglutide 2.4mg vs 2.4% with placebo over 68 weeks in adults with overweight/obesity.
Semaglutide and cardiovascular outcomes in patients with type 2 diabetes (SUSTAIN-6)
Marso SP, Bain SC, Consoli A, et al.. New England Journal of Medicine, 2016.PMID: 27633186
Cardiovascular outcomes trial showing semaglutide reduced MACE by 26% compared to placebo in T2DM patients at high cardiovascular risk.
Semaglutide and cardiovascular outcomes in obesity without diabetes (SELECT)
Lincoff AM, Brown-Frandsen K, Colhoun HM, et al.. New England Journal of Medicine, 2023.PMID: 37952131
Demonstrated 20% reduction in major adverse cardiovascular events with semaglutide in overweight/obese adults without diabetes, leading to new cardiovascular indication.
Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss (STEP 8)
Rubino DM, Greenway FL, Khalid U, et al.. JAMA, 2022.PMID: 35015037
Head-to-head comparison showing semaglutide achieved significantly greater weight loss (-15.8%) than liraglutide (-6.4%) over 68 weeks.
Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide.
Lau J, Bloch P, Schäffer L, Pettersson I, Spetzler J, et al.. Journal of medicinal chemistry, 2015.PMID: 26308095
Davies M, Pieber TR, Hartoft-Nielsen ML, Hansen OKH, Jabbour S, et al.. JAMA, 2017.PMID: 29049653
Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes.
Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, et al.. The New England journal of medicine, 2019.PMID: 31185157
Jiang Z, Tan J, Yuan Y, Shen J, Chen Y. Human & experimental toxicology, 2022.PMID: 36075570
Cimino G, Vaduganathan M, Lombardi CM, Pagnesi M, Vizzardi E, et al.. ESC heart failure, 2024.PMID: 38093506
Kobak KA, Chiao YA. JACC. Basic to translational science, 2023.PMID: 38094694
Beyond Weight Loss: the Emerging Role of Incretin-Based Treatments in Cardiometabolic HFpEF.
Capone F, Nambiar N, Schiattarella GG. Current opinion in cardiology, 2024.PMID: 38294187
Sato R, von Haehling S. Med (New York, N.Y.), 2024.PMID: 38460497
Semaglutide attenuates lipotoxicity-induced cardiac injury by inhibiting Slc27a2 expression.
Pan X, Wang S, Yang X, Jia B, Chen S. Chemico-biological interactions, 2025.PMID: 40456371
The multifaceted effects of semaglutide: exploring its broad therapeutic applications.
Alkhatib M, Almasri N, Alshwayyat S, Almahariq H, Hammadeh BM, et al.. Future science OA, 2025.PMID: 40904035
Stefano GB, Büttiker P, Weissenberger S, Raboch J, Anders M. Frontiers in neuroendocrinology, 2025.PMID: 41047006
AbuAlrob MA, Itbaisha A, Abujwaid YK, Abulehia A, Hussein A, et al.. Journal of Alzheimer's disease reports, 2025.PMID: 41122341
Zheng M, Zhang Q, Siebert HC, Loers G, Wen M, et al.. Experimental neurology, 2026.PMID: 41173224
Griaznova EA. Farmatsiia, 1975.PMID: 5316
50 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
50
Total Trials
12
Recruiting
5
Active
21
Completed
Sponsor: Gilead Sciences · Completed: 2024-12-09
Sponsor: Memorial Sloan Kettering Cancer Center · Completed: 2027-12
Sponsor: Novo Nordisk A/S · Completed: 2019-04-15
Sponsor: Eli Lilly and Company · Completed: 2027-01
Sponsor: McGill University Health Centre/Research Institute of the McGill University Health Centre · Completed: 2027-01
Research Library — 25,528 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
2018 ESC/ESH Guidelines for the management of arterial hypertension
Bryan Williams, Giuseppe Mancia, Wilko Spiering, et al. · European Heart Journal
Research by Bryan Williams et al., published in European Heart Journal. Not conducted by MedTech Research Group.
Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes
Steven P. Marso, Stephen C. Bain, Agostino Consoli, et al. · New England Journal of Medicine
Research by Steven P. Marso et al., published in New England Journal of Medicine. Not conducted by MedTech Research Group.
2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD
Francesco Cosentino, Peter J Grant, Victor Aboyans, et al. · European Heart Journal
Research by Francesco Cosentino et al., published in European Heart Journal. Not conducted by MedTech Research Group.
Once-Weekly Semaglutide in Adults with Overweight or Obesity
John Wilding, Rachel L. Batterham, Salvatore Calanna, et al. · New England Journal of Medicine
Research by John Wilding et al., published in New England Journal of Medicine. Not conducted by MedTech Research Group.
Management of Hyperglycemia in Type 2 Diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD)
Melanie J. Davies, David A. D’Alessio, Judith Fradkin, et al. · Diabetes Care
Research by Melanie J. Davies et al., published in Diabetes Care. Not conducted by MedTech Research Group.
Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes
A. Michael Lincoff, Kirstine Brown‐Frandsen, Helen M. Colhoun, et al. · New England Journal of Medicine
Research by A. Michael Lincoff et al., published in New England Journal of Medicine. Not conducted by MedTech Research Group.
Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007–2017
Thomas R. Einarson, Annabel Acs, Craig Ludwig, et al. · Cardiovascular Diabetology
Research by Thomas R. Einarson et al., published in Cardiovascular Diabetology. Not conducted by MedTech Research Group.
Effects of Once-Weekly Exenatide on Cardiovascular Outcomes in Type 2 Diabetes
Rury R. Holman, M. Angelyn Bethel, Robert J. Mentz, et al. · New England Journal of Medicine
Research by Rury R. Holman et al., published in New England Journal of Medicine. Not conducted by MedTech Research Group.
AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease
Mary E. Rinella, Brent A. Neuschwander‐Tetri, Mohammad Shadab Siddiqui, et al. · Hepatology
Research by Mary E. Rinella et al., published in Hepatology. Not conducted by MedTech Research Group.
The gut-liver axis in liver disease: Pathophysiological basis for therapy
Agustı́n Albillos, Andrea De Gottardi, María Rescigno · Journal of Hepatology
Research by Agustı́n Albillos et al., published in Journal of Hepatology. Not conducted by MedTech Research Group.