SLU-PP-332
SLU-PP-332 (ERR Alpha/Gamma Agonist)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Weight / Metabolic |
| Subcategory | Exercise Mimetic |
| Pharmacological Class | Small Molecule (not a peptide) |
| Subclass | Estrogen-Related Receptor Alpha/Gamma (ERR-alpha/ERR-gamma) Agonist |
| Molecular Type | Small molecule compound developed at Saint Louis University (SLU) |
| Origin | Synthetic — developed by Dr. Thomas Bhurrows's lab at Saint Louis University, published in 2023 |
| Regulatory Status | Research Use Only. Preclinical stage. Not FDA-approved. Published in scientific literature (2023). |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water or appropriate solvent |
| Storage | Refrigerate (2-8°C); protect from light |
Chemical Properties
| Molecular Formula | C18H14N2O2 |
| Molecular Weight | 290.3 g/mol |
| Exact Mass | 290.105527694 Da |
| InChI Key | RNZIMBFHRXYRLL-XDHOZWIPSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
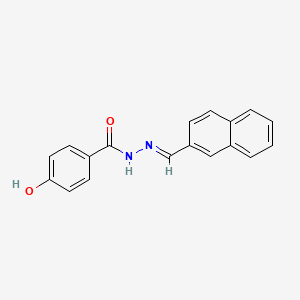
2D structure diagram from NCBI PubChem. This is the actual molecular structure of SLU-PP-332.
Description
SLU-PP-332 is a small molecule exercise mimetic — a compound that activates the same metabolic pathways triggered by physical exercise, without the physical activity itself. Like 5-amino-1MQ, it is not a peptide but is included in the catalog for its metabolic relevance. SLU-PP-332 was developed at Saint Louis University (hence "SLU") and acts as an agonist of the estrogen-related receptors alpha and gamma (ERRα and ERRγ). Despite their name, ERRs do not bind estrogen — they are orphan nuclear receptors (no identified endogenous ligand) that regulate the expression of genes involved in mitochondrial biogenesis, oxidative phosphorylation, fatty acid oxidation, and the metabolic adaptations that occur with exercise training.
When you exercise, particularly endurance exercise, the downstream signaling (through PGC-1α, AMPK, and other exercise-responsive pathways) converges on ERR activation to drive the transcriptional program of metabolic adaptation: increased mitochondrial number and function, enhanced fatty acid oxidation capacity, improved glucose handling, fiber-type switching toward oxidative (type I) fibers, and increased exercise endurance. SLU-PP-332 directly activates ERRα and ERRγ, bypassing the upstream exercise signals and directly engaging the transcriptional program.
In preclinical studies published in 2023, SLU-PP-332 treatment in mice produced increases in running endurance (up to 50% improvement), shifts in muscle fiber composition toward fatigue-resistant oxidative fibers, resistance to diet-induced obesity, and improved metabolic profiles — all without any exercise training. The mice literally became more fit and lean while remaining sedentary.
Clinical Context
SLU-PP-332 is at the cutting edge of exercise mimetic research — a field that aims to pharmacologically reproduce the metabolic benefits of exercise for patients who cannot exercise (mobility limitations, severe obesity, heart failure, post-surgical recovery, frailty). While earlier exercise mimetics like AICAR (also in this catalog) targeted AMPK, SLU-PP-332 targets the downstream transcriptional effectors (ERRs), which may provide more comprehensive and sustained metabolic adaptation. The compound is very new (2023 publication) and is in early preclinical stages.
- NOT a peptide — small molecule ERR agonist; included for metabolic relevance
- Very early stage (2023 publication, preclinical only) — no human data
- Exercise mimetic = reproduces metabolic effects of exercise without physical activity
- Does not replace the non-metabolic benefits of exercise (cardiovascular conditioning, bone loading, psychological benefits)
- ERRs are orphan nuclear receptors — not related to estrogen receptors despite the similar name
- No estrogen-like hormonal effects
- Preclinical safety profile is favorable but human safety is unknown
- Single SKU (5mg, $50.17) — limited supply reflects the novelty of the compound
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to SLU-PP-332
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Research citations are being compiled for this compound.
Check back soon — our team is curating peer-reviewed sources.
Research Library — 73 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Emerging Targets in Type 2 Diabetes and Diabetic Complications
Sevgican Demir, Peter P. Nawroth, Stephan Herzig, et al. · Advanced Science
Research by Sevgican Demir et al., published in Advanced Science. Not conducted by MedTech Research Group.
Understanding, recognizing, and managing toxicities of targeted anticancer therapies
Grace K. Dy, Alex A. Adjei · CA A Cancer Journal for Clinicians
Research by Grace K. Dy et al., published in CA A Cancer Journal for Clinicians. Not conducted by MedTech Research Group.
Beyond adiponectin and leptin: adipose tissue-derived mediators of inter-organ communication
Jan‐Bernd Funcke, Philipp E. Scherer · Journal of Lipid Research
Research by Jan‐Bernd Funcke et al., published in Journal of Lipid Research. Not conducted by MedTech Research Group.
Gossypol Toxicity from Cottonseed Products
Ivana Cristina Nunes Gadelha, N.B.S. Fonseca, Sı́lvia Catarina Salgado Oloris, et al. · The Scientific World JOURNAL
Research by Ivana Cristina Nunes Gadelha et al., published in The Scientific World JOURNAL. Not conducted by MedTech Research Group.
The Role of Vascular Smooth Muscle Cells in Arterial Remodeling: Focus on Calcification-Related Processes
Armand M. G. Jaminon, Koen D. Reesink, Abraham A. Kroon, et al. · International Journal of Molecular Sciences
Research by Armand M. G. Jaminon et al., published in International Journal of Molecular Sciences. Not conducted by MedTech Research Group.
Functions of the aryl hydrocarbon receptor (AHR) beyond the canonical AHR/ARNT signaling pathway
Natalie C. Sondermann, Sonja Faßbender, Frederick Hartung, et al. · Biochemical Pharmacology
Research by Natalie C. Sondermann et al., published in Biochemical Pharmacology. Not conducted by MedTech Research Group.
What can inactivity (in its various forms) reveal about affective states in non-human animals? A review
Carole Fureix, Rebecca K. Meagher · Applied Animal Behaviour Science
Research by Carole Fureix et al., published in Applied Animal Behaviour Science. Not conducted by MedTech Research Group.
A Review of the Modification Strategies of the Nature Inspired Algorithms for Feature Selection Problem
Ruba Abu Khurma, Ibrahim Aljarah, Ahmad Sharieh, et al. · Mathematics
Research by Ruba Abu Khurma et al., published in Mathematics. Not conducted by MedTech Research Group.
An Overview of Current Knowledge on the Properties, Synthesis and Applications of Quaternary Chitosan Derivatives
Emanuelle Dantas de Freitas, Celso Fidelis de Moura, Jonas Kerwald, et al. · Polymers
Research by Emanuelle Dantas de Freitas et al., published in Polymers. Not conducted by MedTech Research Group.
Pharmacology of Free Fatty Acid Receptors and Their Allosteric Modulators
Manuel Grundmann, Eckhard Bender, Jens Schamberger, et al. · International Journal of Molecular Sciences
Research by Manuel Grundmann et al., published in International Journal of Molecular Sciences. Not conducted by MedTech Research Group.